Ether, Enflurane and Explosions “barely for the FRCA”
29 December 2025
Contents
Ether (or perhaps Aether) was first distilled around 1540! by Valerius Cordus, with some jiggery pokery involving gourds, wine, sulphuric acid and some clever distillation.
Enflurane, devised in 1963 subsequently superseded by better agents (as was Ether….)
Explosions, seemingly as old as time itself, but became a concern to anaesthetists, around the time anaesthetists came into true existence (perhaps there was the odd purveyor and or sub-specialty consultant in inebriation about the place before then)
Note, there are two commonly utilised ‘Ethers’ in history for anaesthesia, Di-Ethyl Ether (the subject of this episode) and Di-Vinyl Ether, which we won’t get into, so, if I’m writing ether I intend the former.
Di-ethyl ether acts via GABA-A receptor potentiation — the same mechanism as modern volatile anaesthetic agents — but its blood:gas partition coefficient of 12 makes induction and emergence far slower than sevoflurane or desflurane.
Di-Ethyl Ether Pharmacology Data
| Name | Ether |
| Class | Ether anaesthetic agent |
| Chemical Make Up | (CH3CH2)2O |
| History | Ether first use as a surgical anaesthetic was by a GP Dr. Crawford W. Long on March 30, 1842, in Georgia, used to remove a neck tumour from a boy, James Venable, Dr Long’s use was understandably not noticed by the public, he didn’t publish, and Twitter was yet to exist. (he had been using the Ether for a laugh or two with the local folks where he noticed people sustaining non painful injuries) William T.G. Morton on the Night of September 30, 1846 extracted a tooth from Ebenezer Hopkins Frost, if anyone has had a severe toothache you can appreciate you would give anything a go, one of several apparent suggested ‘we were first’ anaesthetics. 4 years after Dr Crawfords neck rummaging, it was deemed a smart move to publicly demonstrate ether anaesthesia to the world. This happened at Massachusetts General Hospital -in Boston on October 16, 1846, where a mass under a jaw was removed from a Mr Gilbert Abbott, with Mr Frost vouching for the Ether. Hence Ether day on October 16th. Morton’s public demonstration of painless surgery sparked rapid adoption of ether. Prior to this use, Ether had been administered via teapots in a clinic in Clifton, Bristol, UK. for general ailments, and nitrous oxide was also noted at the time to be an amusing pneumatic to inhale. It sounded like they may have accidentally anaesthetised one fella and ‘considerable fears were entertained for his life’ but the individuals at the time did not make the cerebral step towards surgery! To much Ether addles the mind? |
| Isomer Status | |
| Colour/Appearance | Clear colourless liquid – Flammable and Explosive |
| Stability | |
| Molecular weight | 74g/mol |
How to make Ether in a Snap
Mix sulphuric acid with a nice bottle of Pinot Noir
Hollow out a gourd or two for fractional distillation
Don’t miss any important steps
Obtain Ether, store in sealed teapot
| Ether Boiling Point | 35 °C |
| Ether Saturated Vapour Pressure (@20 °C) | 56.7 kPa |
| Mac of Ether | 1.92% |
| Blood:Gas Solubility Coefficient Ether | 12 |
| Oil:Gas Solubility Coefficient Ether | 65 |
| Flammable Ether Mix | In AIR as little as 1.83% v/v (up to 48%) will deflagrate |
| Explosive Ether Mix | In OXYGEN 2-82% Ether v/v will go boom. |
Ether PharmacoDynamics
Ether produces general anaesthesia via positive modulation of the GABA-A receptor throughout the CNS, with a notably high blood:gas partition coefficient of 12 — producing slow induction and prolonged emergence compared to modern agents.
| Mechanism of Action | Ether works like all other volatile anaesthetic agents, with modulation of the GABAA receptor throughout the CNS, |
| Chief Effect / Actions | Hypnotic |
| Dose | See End Tidal Mac for Dosing. – Ether Vapourisers would deliver 3-20% concentrations. |
| Side Effect Profile | |
| Cardio-Vascular Side Effects | Chronotropy : Ether has a positive chronotropic effect (vagolysis and SNS stimulus) Inotropy : Negative in vitro, but with the SNS activation this isn’t seen in vivo Lusitropy Coronaries: Vasodilating Vascular Resistance: Increased (SNS activation) in deep planes of anaesthesia, with a vasodilatory effect in lighter planes CO increased by around 20% No catecholamine sensitisation |
| Respiratory Side Effects | Rate – Increased Depth – Decreased Parenchymal effects: > Hypoxic pulmonary vasoconstriction inhibited > Bronchodilating > Irritant = Coughing++ (and breath holding) especially if dose increased too rapidly |
| Central Nervous System Side Effects | ICP : Dose Dependent Rise Seizure Threshold : Unaltered, but patients can develop clonus in lighter planes Cerebral Vasculature: Ether is a cerebral vasodilator CMRO₂ Nausea and vomiting in 50% of patients! Analgaesic Effects! Progressive pupil dilation Hypersalivation – hence the historic atropine premed |
| Metabolic Side Effects | Ether stimulates gluconeogenesis, and may also cause a Type B lactic acidosis state (they didn’t have metformin then to test it properly I suppose!) |
| Gastro-Intestinal Side Effects | nil |
| Genito-Urinary Effects | Ether causes renal artery vasoconstriction |
| MSK Side Effects | Potentiates NMB drugs |
| Obstetric | Uterine hypotony |
| Toxicity Signs | Wide safety margin given how long it takes to get drug into the patient, which was convenient given they originally sloshed it on to a sponge. |
Ether Pharmacokinetics
Ether is eliminated 97% via the lungs; only 2–3% undergoes hepatic Phase I metabolism to acetaldehyde, alcohol, and acetic acid, with negligible renal excretion.
| Absorption | Note that Ether is massively soluble in blood, it is 17x more soluble than [sevoflurane](/episode/sevoflurane/). That means it takes quite some time to get to an effective concentration. |
| Distribution | Extensively, favouring areas receiving higher blood flows |
| Metabolism | Liver 2-3% of Ether undergoes liver metabolism Phase I : Phase II : Acetylaldehyde, alcohol, acetic acid and CO2 are produced. Remember: Phase I : [oxidation, reduction, hydrolysis] (more cytochrome action here, more O2 required) Phase II : [conjugation, glucoronidation, acetylation, sulphylation] |
Active Metabolites: Other Routes: | |
| Elimination | 97% excreted via Lungs |
| Other Notable Information |
Enflurane Pharmacology
The most important thing to know about Enflurane for the FRCA exam, is that it is a structural isomer of isoflurane.
- It is a clear and colourless, non flammable liquid, anaesthesia is at 1.6% MAC
- CVS effects of Enflurane include a tachycardia, negative inotropy and coronary and general artery vasodilation
- RS effects include, Bronchodilation and significant Resp depression
- CNS wise, Enflurane can induce seizures, and increases ICP and cerebral blood flow.
- It is 2.4% metabolised in the liver,
- A further 2.4% is excreted via urine, sweat, faeces and the rest via exhalation.
See MAC and Anaesthetic Depth for the full significance of MAC values in clinical practice.
Through the Fire and Flames, and on to Explosions.
We’re going to go through managing an airway fire. Then we shall talk about explosions.
Why? Because there is an BJA article that was probably quite fun to write that handles explosions, I’ll link to it in the references. Also because I found a book called Physics for the Anaesthetist, including a section on explosions in a charity shop a number of years ago . It actually transpires that when I’ve looked at this book properly, it was written by Sir Robert Macintosh and some colleagues. Macintosh of MacBlade Fame + all his other exploits at the time
Fortunately, we live in a world where in-theatre explosions are much less likely. This is because, as we’ve mentioned earlier in the show, there aren’t as many explosive gases knocking around and safety practices are fairly maximal.
Tis certainly important for us to know about how to deal with a fire. That could be an airway fire, or it could be a fire on the patient.
If the surgeons have been a bit liberal with their chlorhexidine, failing to mop it up, or failed to allow it to fully evaporate, there is a problem. The alcohol content of chlorhexidine antiseptic is really high, you can end up with an invisible fire because of the nature of the flame, alcohol emits little visible light spectrum when combusting.
And I think it’s quite interesting to understand a bit more about what an explosion is. We need to define or die a number of things and appreciate how flames behave through flammable materials in order to differentiate What? What a flame is, what deflagration is, and what an explosion is.
Definitions or Death
Flame – a hot glowing body of ignited gas that is generated by something on fire – combustions that produce light cause a flame
Cold Flame Reactions – An oxidation reaction of a mixture that will only sustain whilst there is a source of heat energy supplied.
Deflagration – burn or cause to burn away with a sudden flame and rapid, sharp combustion (in a controlled system)
Conflagration – an extensive fire which destroys a great deal of land or property, think wild fire, (con- as a prefix means with, or thoroughly)
Combustion – a rapid, exothermic redox reaction (oxidation) between a fuel and an oxidant, usually oxygen, that releases significant energy as heat and light, often producing oxides, carbon dioxide, and water as products.
Flash Point – A property of a liquid fuel, whereby the temperature of a volatile liquid fuel will give a flash of flame across its surface when a flame is passed across it. Imagine, very hot petrol, doing lots of vapourising, would probably burn raucously. Whereas very cold petrol, might only vaporise a little, and have a more sedentary flame skip across its surface.
Explosion – Really anything that achieves a volume of expanding gas in very short time frames. The shorter the time frame, the more boom there is.
Sub Types of Explosion
- Mechanical – Over loading something with compressed air, if you heated a VIE and closed the release valves you would eventually cause a boom as all that liquid o2 wants to be a gas 861x the size
- Nuclear – rapid release of huge quantities of energy causing heat expansion of the gas around the explosion alongside all the rest of it.
- Chemical (detonating types like TNT/’C4’/Dynamite AKA High Explosives) and deflagrating types, so called low explosives)
Shock Wave – a sharp change of pressure in a narrow region travelling through a medium, especially air, caused by explosion or by a body moving faster than sound waves through that medium
Detonation – combustion of a substance which is initiated suddenly and propagates extremely rapidly, giving rise to a shock wave.
For a Fire to Get 11A* at GCSE it generally requires the right combination of:
Heat source
A spark, or a flame or sufficient compression of a gas. Or anything that delivers concentrated heat (a magnifying glass, Laser, Electrocautery) or in true NHS fashion, everyone knows of a slightly dodgy electrical socket with some tape over it saying do not use, nor should you toast bread using open flame or modern toasting devises as these are notoriously sparky and will be sufficient for deflagrations.
Oxidisers
Generally this is oxygen, as a fraction in air. It can also, in the anaesthesia setting be nitrous oxide, nitric oxide of hydrogen peroxide.
Fuel
Material that contains molecules that will reconfigure if oxidised into a lower energy state – releasing heat energy.
Peri-operative fuel sources include bowel gas, surgical prep, drapes that melt, burn and cook when the laparoscope that’s been set to 9000 lumens are left pointing at them and probably many other things.
Mitigating Fire Risk
The less exciting but still very important thing here, is tactics to mitigate the potential for fire.
- Be as cheap as you can with oxygen, and don’t leave a face mask blowing under the blankets in Resus, nasal specs under the sheets is another thing, and static pings from a NHS blanket is not unheard of.
- Unplug the nitrous from the anaesthetic machine and throw the piping out, (or if feeling less maniacal / extreme, just don’t use nitrous.)
- Fire safety plan when using lasers,
- Keeping an eye on the sloshing of high-alcohol solutions.
- Being cautious with igniting body hair.
- Regular PAT testing of electrical kit, anything that you touch that tingles needs to be turned off. Isolated from any staff thinking about switching it on again
- Don’t grease your BODOK seals, even if there is a tank/yoke leak. The compression effect of tank pressure on the greased seal can lead to flames from the back of your anaesthetic machine,
- however cool it may sound to have a backfiring anaesthetic machine, it is in fact very hot and quite precarious, and may ignite all the other stuff dangling on the back of the machine.
For more fire safety learning, do attend induction, and check out the BJA article, as whilst I do jest, fire is exceedingly, devastatingly bad in a hospital packed densely with unwell people who cant evacuate in an orderly manner.
Handling An Airway Fire
First step is actually spotting one, there may be a flash, a pop or a funky smell, +/- smoke
Second step is hollering FIRE
And in a co-ordinated manner:
- Stop Ventilating,
- Deflate if possible and remove airway devices
- Slosh copious saline into the airway – a jug of water must always be available in surgeries with fire risk
- Surgeon should remove any remaining debris once saline is in (any plastic will have cooled down)
- Ventilation should be resumed, with recommendations to use low fiO2 fractions or room air via an AMBU bag.
> If the fire is instead not contained and spreading over the patient, then a CO2 extinguisher, should be used if safe and a fire alarm certainly sounded.
If control was attained, then the airways need to be inspected for injuries, (bronchoscopy) further airway toilet as needed, and a plan formulated. There is potential for airway and lung parenchymal injury, and as such, keeping the patient sedated and intubated for a period of observation is necessary to observe for ARDS / gas exchange complications.
Sources of Ignition
Autoxidation
Once upon a time, Ether explosions came about due to chemical instability introduced by exposure to light and air, whereby peroxides (hydrogen peroxide for example) were formed in the solution, this situation is very labile, and act like detonators in high explosive mixes (High explosives often need fairly significant energy to trigger their reaction)
This ‘self’ creation of a detonator is called autoxidation, and accidental shaking of a sample of ether exposed to light in a lab has led to explosions, introducing anti-oxidants to your ether solution, and storing it in the dark, mitigates this.
Spontaneous Ignition
If a particular gas mix, is heated to a particular temperature, it will undergo simultaneous combustion throughout the whole mixture.
Somes gases burn simply when warm enough, others if allowed to shift towards room temperature will spontaneously combust. Highly precarious things, thankfully there are very few that do this. All gas mixes generally have an auto-ignition temperature.
As you would expect, most things don’t spontaneously erupt into flames at general atmospheric temps, because otherwise our planet would be much less likely to support life as we know it!
Wood will auto ignite at 300 °C and isopropyl alcohol at 399 °C
Sources of Local ignition – more relevant to the jobbing gas slingers of the modern age
- Open Flames – there are very few theatres which now have an open coal fire for warmth
- Cigarettes ……
- Laparoscopes with their high intensity light sources burning drapes (seen it)
- Diathermy
- Arcs from faulty switches
- Static build ups – only occur in non-earthed or poorly conductive materials which are unable to dissipate their -ve charge.
Mitigations for Ignition sources
- Maintaining a degree of humidity, leads to surfaces being a tad moist, bolstering their conductivity and hence static potential differences dissipate before they can be come dangerously charged.
- Using materials that are more conductive, ie, rubber that is altered to be a less effective electrical insulator.
Explosives in Anaesthesia
The explosions that were seen in anaesthesia often weren’t the absolute fastest, like dynamite / TNT. Instead tending to be a mixture that might be burning its way through a mix journeying along a tube, vide infra.
We should certainly make a distinction between mixes with air and oxygen. Ether plus air does not explode very much at all [1.83% v/v (up to 48%) will deflagrate], whereas ether and oxygen has an explosive or detonating range of between 3 and 33% ether oxygen mixtures.
There have been a few other explosives used in and around anaesthesia:
- The original ether, aka diethyl ether, had a friend called di-vinyl ether, which was more boomy and combustible again.
- Ethyl chloride. which is used for cold sprays testing spinals in places that haven’t just progressed to a bit of metal on a stick in a fridge. Ethyl chloride in oxygen produces an explosive mixture with a range of 4 to 67%, which is broad. Whereas in air… 3.8 to 15% concentrations will explode.
- Cyclopropane, which was once upon a time used, Mixed with oxygen, a range of 2.5 to 60% cyclopropane to oxygen would result in booms and deflagrations, whereas cyclopropane and air will only deflagrate.
- Nitrous oxide supports combustion and it burns hotter compared to a solitary oxygen mix.
- As the nitrous in a mix is heated, the oxygen is liberated, providing more than the 21% available in air.
- Hence its use to jazz up engines in need for speed underground 2
Combustion Reactions for the Jobbing anaesthetist
**A representation of a simple combustion reaction would look like:
**
Reactants (Gases A + B) >>> Heat Added >> Chemical rearrangement >> Products (Gases C+D etc) +/- Heat +/- Photons
Generally, you need to deliver energy to break a bond, when you make a bond, further energy is released.
This is the activation energy of a reaction, which needs to be added to the system for a conformational change in the molecules of that system to occur leading to new chemical arrangements that will either release or absorb energy, depending on the nature of the reaction.
And in most systems, these reactions will go from a less stable to a more stable set of molecules when they oxidise i.e. from more reactive to less reactive.
An Example of Combustion we are all familiar with
A better example of this is wood. When you burn wood you’re not just burning the cellulose of the cells of the wood but the saps, the sugars etc. These all burn at different temperatures. There are varying amounts of it within it and often they’re shifted into their volatile or gas phase before being ignited.
If your fire lacks heat energy, you’ll often see a lot more smoke, and that’s because you are incompletely combusting the generated gases and vapours of volatile things coming out of the wood. Hence why smokey grotty. Whereas if you can get sufficient heat in your fire, you end up with much cleaner smoke-free flame as everything is combusted. As you might expect, if you successfully and entirely combust all of the material, you will generate more heat as opposed to wasting stuff.
That’s why wood-burning stoves are designed in a manner that A contains the heat, B will provide sufficient oxygen to the materials in order to combust them. Poorly combusted materials in your home. The wood-burning stove furs up your flue or chimney with non-combusted but combustible materials. If you then go and actually get a rip-roaring fire going in there, you will ignite those materials now deposited up your flue. Remember they deposit as they cool, they’re more likely to stick to the flue. And therefore you can get a flue fire.
The heat released from a reaction will go on and increase the rate of that reaction as long as the heat is contained and isn’t being freely dissipated.
Factors Influencing Reaction Rates
- Fuel Mix
- Heat gain / Loss in a system
- Physical environment enclosing (or not enclosing) the reaction.
Fuel Mixtures
In a perfect world 10 x A + 10 x B >>>>> 10 x C and 10 x D + Heat + Light
This would be a stoichiometric mixture. Its reaction rate would not be constrained by the available substrates for the reaction.
But if you swing to either side of this stoichiometric mix, you either end up with a rich mixture or a lean mixture. You might have come across this if any of you folks are motorbike aficionados. You can fiddle around with your carburettor and provide your engine with more or less fuel.
The more you swing towards leaner or richer mixtures, the slower the rate of reaction will become, noting again that the stoichiometric mixture is the most rapid to react.
Now, depending on what you have mixed in this tube dictates that reaction speed, and whether or not the heat generated by the reaction is sufficient to maintain that wave front of flame.
Heat Gain / loss
As a rule of thumb- An increase in 10 °C doubles reaction rates.
If losses are too high, then you have an imbalance that leads to failure of propagation of that burning wave front through your tube. Vide infra
- If there are non-combustible gases in this system, those gases will get heated unnecessarily. For example, if you’ve got nitrogen in this tube as well. its inert, and not involved in the reaction, but it’s going to soak up some of the heat generated,
- Heat may be conducted or radiated out depending on the material the system is made of.
- If the gas is humid, i.e. there’s water vapour again, that’s another thing that can get heated.
- And equally the gases of combustion, i.e. the resul tant reactant products themselves, are going to be warmed, but not useful for further reactions.
Reaction Vessel
Consider an explosion within a room, or a fire within a room, that room gets hotter and hotter and hotter, speeding up and speeding up that reaction. as long as the other two elements required fuel and oxidizer remain present in that system.
Reaction rates within these systems are often described like half times, i.e. how long does it take for half of the reaction to occur?
And these rates of reaction aren’t purely dependent on heat available, but also the mixture of fuel and oxidizer in the system against things that don’t get involved. Nitrogen is inert, so nitrogen in air does not get involved in combustion. If you remove that nitrogen from the system, your ratios of agent that oxidises versus agent that does the oxidizing may lead to faster reaction times.
The Behaviour of a Combusting ‘wave front’ within a system
As anaesthetists, we are somewhat interested in deflagrations and explosions that might happen within our ventilatory apparatus. i.e. in tubes.
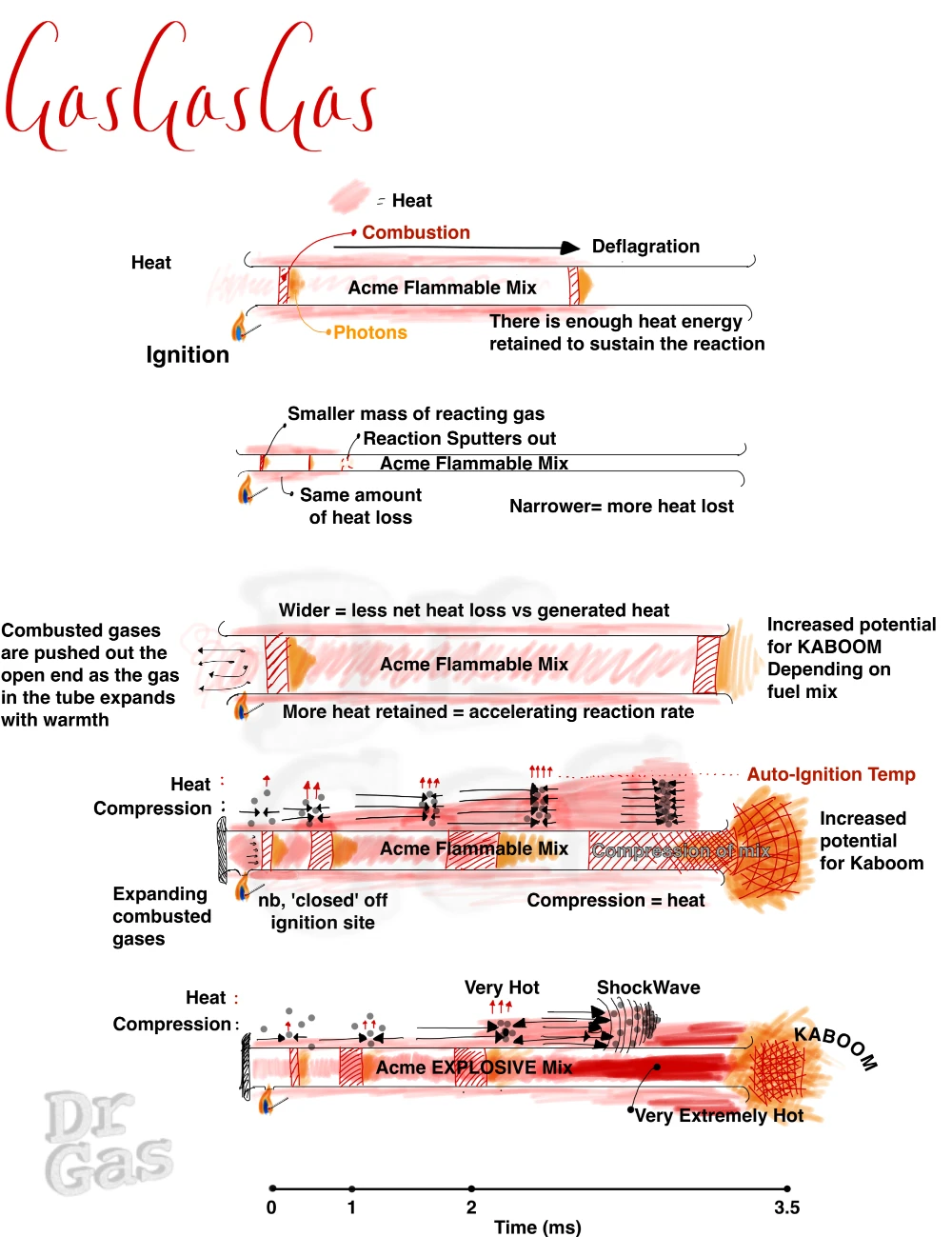
Imagine an open ended tube, with a flammable gas mixture within it. We introduce a heat source at one end (A).
Four outcomes may occur
- The flame sputters and fails as there is insufficient heat to maintain combustion
- The flame tracks from A though to B speeding up a bit as it goes
- The ignition near immediately causes a burst of flame and heat from each end of the open tube
- For the astute, a fourth option would be, the mix fails to ignite
As long as that mix can self-maintain its combusion(reaction) with the heat it generates, you will end up with a wave of flame transiting that tube from one side to the other, from A to B.
If you were to start the reation from the middle, it would propagate in both directions,
The speed at which it transits from A to B is directly related to the speed of the reaction.
The flame that you see in this tube is that wave front where the oxidation reaction is actually taking place, generating photons.
click to enlarge
What about different tubes?
We need to think about a few different permutations of this tube. My original tube has a hole at both ends. But what if you have a closed end and you start your ignition from that closed internal end, the nature of the expansion of those gases and the heat agenerated will go in a single direction, it doesn’t escape out in both directions. This can lead to an expanding and speeding up wavefront as you heat the reacting . If your tube is in a vertical position and you light it from the top, The propagation of that flame downwards is quite slow, as a lot of heat is lost out of the top. Whereas if you start that fire at the bottom, you have a much more rapid reaction rate.
So, now we know that if we start a fire in a tube, it propagates along that tube. We know that there are rich mixtures, lean mixtures, and stoichiometric mixtures. And by that virtue, we need to consider that at some point, with the correct conditions, our propagating flame (deflagrating) down our tube will turn into an explosion down our tube. This is generally when we are in that middle stoichiometric ground, with a mix that can react fast enough to tip into detonation. Bear in mind that an O2:fuel mix has a much broader range of boom than an air:fuel mix, noting, only some air fuel mixes will detonate.
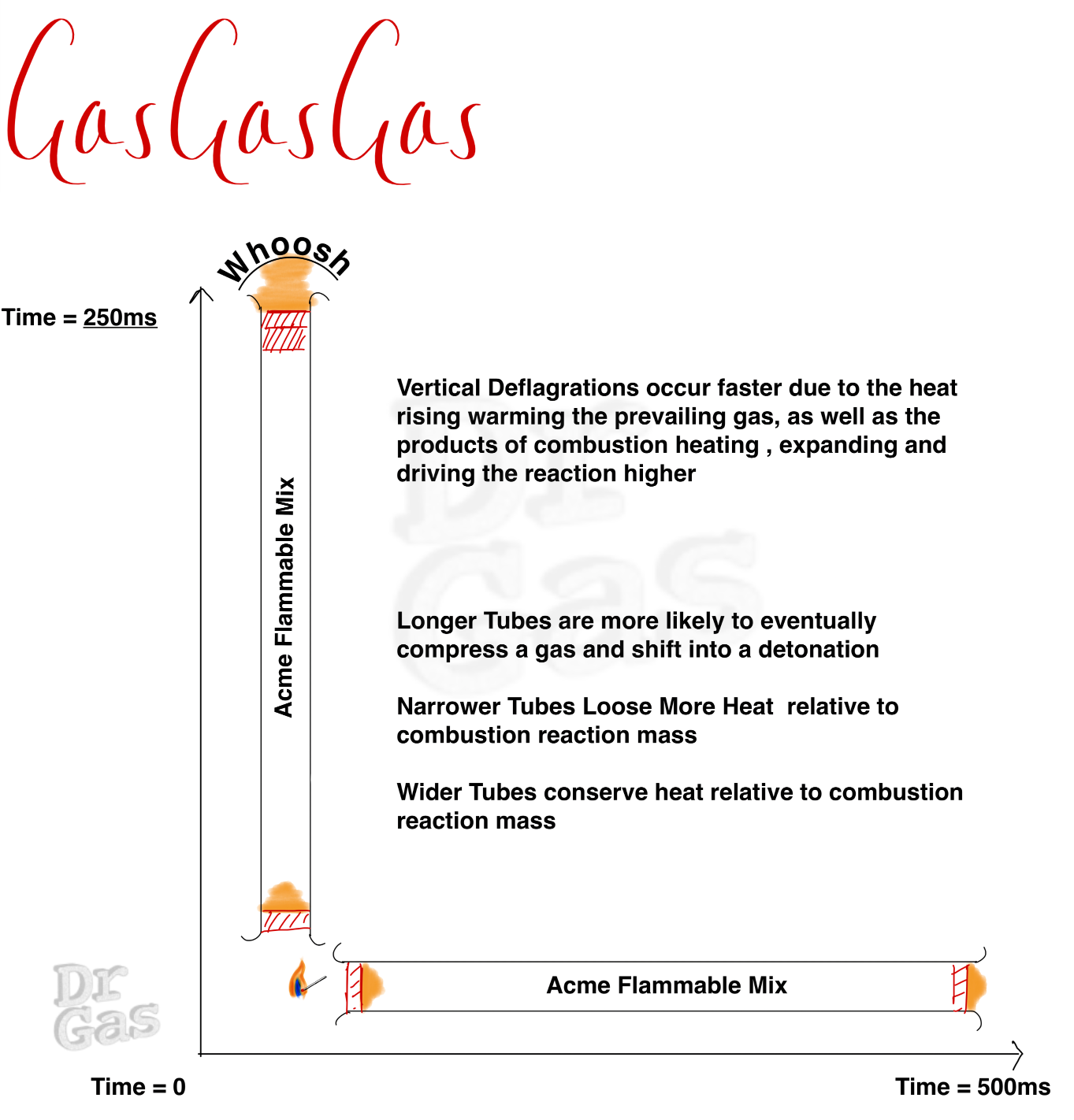
click to enlarge – Note the TIMES! vertical is faster!
Detonations
Now, imagine that we have a flame propagating down a tube and it’s going faster and faster and faster, getting hotter and hotter. It starts to compress the air ahead of it. That act of compression adds further heat, (we know if you increase pressure in a system, the heat goes up i.e you make your bicycle tyre pump heat up as you use it by repeatedly compressing the air until it hits a threshold of a valve which releases into the tyre.
This speeding up reaction will eventually trigger a rapidly expanding wavefront of combustion, compressing gases ahead of it, this is called a detonation, where fast combustion is propagated by a shockwave. These shockwaves are actually supersonic down our tube. The reaction will then occur at a rate of 2 to 3 kilometres per second, hitting really high temperatures like 2,500 to 3,500 °C, and achieving up to 30 atmospheres of pressure. So that then leads us to say that a detonation is fast and a deflagration is slow.
Hand on you said deflagration? Remember that’s when you light your gas mix at one end of the tube and it somewhat slowly propagates along the tube to the other end.
Detonations have many factors that influence them, and the nature of our tube is once such thing. if it was a wide and a long tube, it would foster a faster reaction.
Implications of fire in a breathing circuit
Deflagrations in a circuit may progress all the way to the alveolus, but in modern circuits, this flaming wave font might be held up at the HME with a much moister environment within it and on the opposing side of it, demanding more of the heat energy of the reaction to continue.
Detonation within a breathing circuit doesn’t lead to just airway burns of the patient but that pressure spike in the circuit can lead to blast injuries of the lungs as well much like the concerns we have for soldiers involved in blasts especially in contained places or of significant kinetic energy they can end up with blast lung which is a life-threatening issue that looks a bit like ARDS.
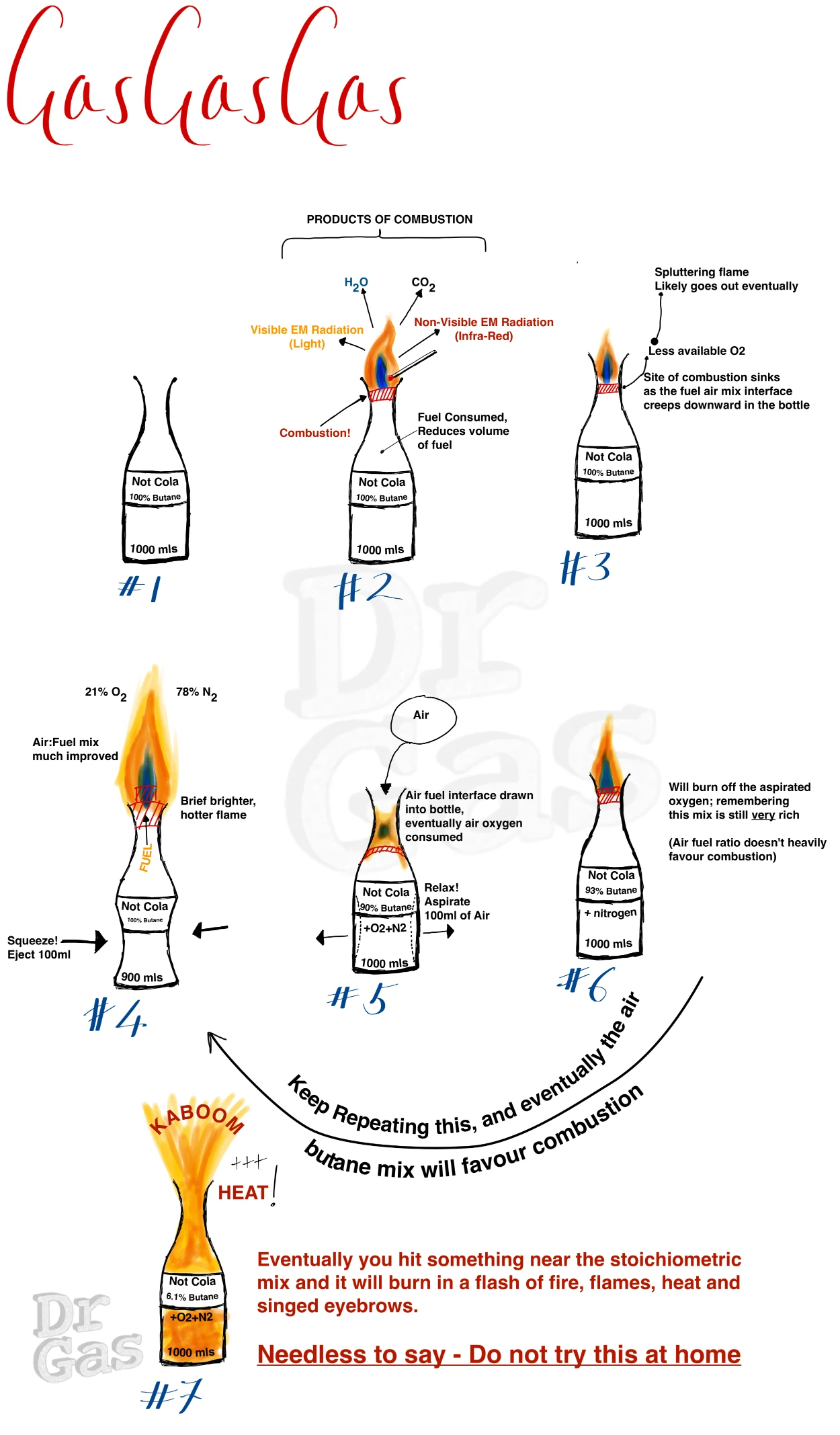
Time for an analogy, because that was a lot of facts. As a young idiot, I took a 1 litre Coca-cola bottle, drank the Coca-Cola, and then filled that Coca-Cola bottle with butane from a lighter.
I then lit that. It burned slowly with a flame from the end, and if I squeezed the bottle and ejected some of the butane, it would puff out a little fireball, which all was fun. Otherwise it went out.
As I was fiddling around trying to re-ignite the mix, I squeezed the bottle and aspirated a load of air inside it. That aspirated air mixed with butane, Creating a now flammable butane:air mixture when before it was just butane with no oxidiser, this system actually blasted flames out of the end of the coke bottle all over my hand and blistered the crap out of my hand.
Naturally, I did not own up to this.
My hand recovered within a week and a half, the blisters burst. Very painful, but demonstrates that that was initially a rich mixture. It was just about burning when some of the butane escaped the end of that bottle mixed itself down into a flammable mixture.
Eventually it hit a sweet spot where I’d inadvertently created a near stoichiometric mixture that went vwoomph in a flash of burning that was much hotter and sizzled me.
After that I was much more careful to point the bottle away from myself, I remained an idiot.
Don’t try it at home folks!
Hope you have enjoyed this, and if you’ve made it to the end, 10 points to Gryffindor!
click to enlarge
References
- Environmental emergencies in theatre and critical care areas: power failure, fire, and explosion Miles, Lachlan F.Scheinkestel, Carlos D.Downey, Glenn O et al. Continuing Education in Anaesthesia, Critical Care and Pain, Volume 15, Issue 2, 78 – 83
- LeVasseur, Ryan M.D.*; Desai, Sukumar P. M.D.†. Ebenezer Hopkins Frost (1824–1866): William T.G. Morton’s First Identified Patient and Why He Was Invited to the Ether Demonstration of October 16, 1846. Anesthesiology 117(2):p 238-242, August 1, 2012. | DOI: 10.1097/ALN.0b013e31825f01b7
- DUNCUM BM. Ether anaesthesia, 1842-1900. Postgrad Med J. 1946;22(252):280-290. doi:10.1136/pgmj.22.252.280
- Auto-ignition temperatures
- Sir Robert Macintosh, Physics for the Anaesthetist, including a section on explosions, 3rd Ed 1963
- (Internet Archived version – https://archive.org/details/physicsforanaest0000sirr/page/n5/mode/2up)
Thanks for listening. Take it day by day, don't overcook yourself — keep studying.
Transcript
58 min listenRead the full transcript
Gas Gas Gas Podcast – Christmas Special
A comprehensive exploration of ether history, enflurane basics, and the science of anaesthetic explosions for FRCA primary exam preparation.
Introduction
00:00-00:56
Summary:
• Welcome to Gas Gas Gas – FRCA primary exam preparation podcast
• Designed for multitasking – gym, commuting, daily activities
• Website: gasgasgas.uk for show notes and references
Please listen carefully. Hello, Team Anesthesia! Welcome to Gas, Gas, Gas. This is the best anaesthetic science podcast for the FRCA primary exam. Our goal is to fill your brain with all this highly useful information. Now, you might be in the gym right now, commuting, or ironing your scrubs, and there’s no judgement here. Gas, Gas, Gas will prime your brain for the monsoon of knowledge you need to imbibe. But regardless, the revision is eventually going to end. But for now, expect facts, concepts, model answers, and the odd tangent. Now remember to check out the website, that’s gasgasgas.uk. There are show notes there with all the detail, plus links to foundational reference papers and anything else useful I find for you guys. Anyway, buckle up, get ready for your mind to be bent into a new shape, and let’s get on with the show. Hi, hello everyone, and welcome to Gas, Gas, Gas. This is a Christmas-themed episode, much like Die Hard as a Christmas movie. brought ether, and enflurane, and explosions to an anaesthetic stable near you. We’ll try and shoehorn as much Christmas cheer into this as possible for those post-Christmas blues before that New Year’s blowout that I’m sure you’re all super looking forward to working. So, we’re going to talk ether, because why the hell not, it’s interesting, we’re not studying over Christmas. We’re going to touch very, very briefly on enfluraine, purely because I needed something that sounded good between ether and
Episode Overview
00:56-02:55
Summary:
• Christmas-themed episode: ether, enflurane, and explosions
• Key reference: Sir Robert MacIntosh’s 1943 textbook
• Balance of FRCA relevance and historical interest
brought ether, and enflurane, and explosions to an anaesthetic stable near you. We’ll try and shoehorn as much Christmas cheer into this as possible for those post-Christmas blues before that New Year’s blowout that I’m sure you’re all super looking forward to working. So, we’re going to talk ether, because why the hell not, it’s interesting, we’re not studying over Christmas. We’re going to touch very, very briefly on enfluraine, purely because I needed something that sounded good between ether and in the title. But enflurane might come up. There might be a question in the exam. But it’s a very limited scope. And then we’re going to chat explosions. I’ve been mentioning an explosions episode for quite some time. Turns out it’s quite a meaty subject. The book that a lot of this knowledge is from, because there’s only a whiff of it on eLearning for Help, and I will absolutely point out that if you’ve got to a point in the exam when they’re asking you about explosions and ether, then somehow you have made. And I’m sure the examiner is sweating under the collar trying to think of the next question to ask. But it’s interesting, it’s curious, and if you end up doing a shimmelbush ether anaesthetic under a bush when the world ends, well, you’ll know how to not blow yourself up and your patient and colleagues. The big shout-out, I suppose, goes to Sir Robert MacIntosh, who was the first professor of anaesthesia at Oxford university in the United Kingdom at the Nuffield. School of Anesthesia down in Oxford land, because it’s all out of this book, and actually it’s a really good book. Although admittedly it uses some imperial vibe unitary, because it was written in 1943, but actually it’s done a really good job of explaining stuff. So if anyone sees it knocking around in a charity shop, grab it. I think I got it for like a pound. A link to the Welcome Library page for it is in the show notes. for anyone who is curious about some old-school anaesthesia text reading, which I’m sure, if you’re listening to me, you probably are. So before I get too stuck into talking to you about what I’m going to talk to you about, I best get into talking to you what I’m going to talk about. That is ether. So I was really quite surprised to find out that ether was first distilled in 1540 by someone called Valerius Cordus, which is a cool name, involving gourds, wine, sulphurus.
History of Ether
02:55-07:23
Summary:
• 1540: First distilled by Valerius Cordus
• 1842: First surgical use by Dr Crawford W Long
• 1846: William T.G. Morton’s public demonstration
• Earlier British experiments in Clifton, Bristol with teapots
for anyone who is curious about some old-school anaesthesia text reading, which I’m sure, if you’re listening to me, you probably are. So before I get too stuck into talking to you about what I’m going to talk to you about, I best get into talking to you what I’m going to talk about. That is ether. So I was really quite surprised to find out that ether was first distilled in 1540 by someone called Valerius Cordus, which is a cool name, involving gourds, wine, sulphurus. and some fairly clever distillation processes. There’s a link to an article that explains his process in great detail if you’re interested. But whilst we distilled it, and when I say we, humanity, distilled it in 1540, no one really knew quite what to do with it. So it was first used as a surgical anaesthetic in 1842, March 30th, in Georgia, United States. by a GP, Dr Crawford W Long. Now, I think GP today and GP in 1842, probably quite different because this chap used it as a surgical anaesthetic to whip out a neck tumour from a boy called James Venable. Now, Dr Long was just sort of cracking on with it and getting on with his tasks and being clever about it and wasn’t perhaps the best to get his work out on Twitter at the time. So he was originally not quite completely credited for this. So ether was actually being used a little bit like nitrous oxide is being used today by kids, adults, anyone huffing up those little ampules of nitrous that they use for whipping cream and having a laugh. Well, they were using ether for a laugh back then. And it spread across the United States fairly quickly. And some local folks were, you know, getting blasted on ether. and then noticing that people were sustaining non-painful injuries. I think Dr. Long also probably partook in passing the ether and noticed bruises that he didn’t recall getting. And that’s where he drew the logical step that it seemed to obliterate painful stimuli. Four years after this, there was a public demonstration that introduced ether to the world, really, by William T.G. Morton at Massachusetts. It’s general hospital in Boston. That was in 1846. And he extracted a tooth from a bloke called Ebenezer Hopkins Frost, which again is a cracking name. Morton’s public demo of this painless surgery sparked the rapid adoption of ether around the world. But, naturally, the British got there first. But by accident, and no one noticed its benefit. So prior to all this, in the Clifton area of Bristol, of all things, in a Hospital of sorts for pneumatic diseases. We were starting to get obsessed with the airs. There’s a humour, I maybe, I don’t know. People were inhaling ether via teapots, which is a very British process, alongside inhaling nitrous oxide via teapots. It was noted at the time an amusing pneumatic to inhale, and it sounded like there was one bloke who went quite hard on the ether, and subsequently was rendered unconscious for about 30 hours by the sounds of it. Now naturally everyone was rather perplexed and there is an entry stating someone had considerable fears were entertained for this man’s life but no one had made the association that actually if you can render someone unconscious for 30 hours maybe you could nick their kidneys or take out a tooth. So that’s the history of ether and where it all cracked off and it has been used for quite some time since then prior to the of other volatile agents. And we will do a volatile agent timeline history type episode at some point in this volatiles chapter. I’m acutely aware that this is not a history of anesthesia podcast just yet, but that you guys need to study for an exam. So naturally we will focus on the stuff you need to know as well. But I’m sure you can have a lovely conversation with the father and mother of your anaesthetic department who’s been there for yonks. Maybe they knew someone who used to use it.
Physical and Chemical Properties of Ether
07:23-09:20
Summary:
• Molecular weight: 74 g/mol; Boiling point: 35 °C
• MAC: 1.92%; Blood-gas solubility: 12; Oil-gas solubility: 65
• Flammable in air: 1.83-48%; Explosive in O₂: 2-82%
So, ether is a clear, colourless liquid that is flammable and explosive. It’s quite a small molecule, 74 grams per mole, and it pretty much looks like an oxygen molecule with two gull wings if you draw it out in an organic chemistry carbon backbone manner. So it’s a CH3CH2202. You definitely don’t need to know that for the exam. It boils at 35 °C, and it’s saturated is about 56 kilopascals. So its saturated vapour pressure is actually quite high. Not quite as high as desflurane, which is in the 80s, but it boils at almost room temperature as well. The MAC of ether is 1.92%, so actually quite a small dose. But its blood gas solubility coefficient is 12. Now remember, sevoflurane’s BG coefficient is 0.69. So that is an absolute truckload of excess ether floating around in the blood not getting involved in tickling those GABA-A receptors. As you’d expect given its MAC of 1.9 you must therefore be able to infer that its oil gas solubility coefficient must convey a high potency and we’re right it has an oil gas solubility of 65. Other important physico-chemical data for ether is something we’re all going to be less familiar with, and that is what mixes of ether with air or oxygen cause ignition or explosion. So in air, if you have 1.83% ether up to 48% ether, it will ignite and it will deflagrate. Deflagration, deflagrate. We’re going to define that later. To create an explosive mix of ether, you need to add oxygen. And at concentrations between 2% and 82%, ether will go boom, kaboom, boom. Much like those penguins from Madagascar who like to blow stuff up. Ether, maybe they could have used it. So pharmacodynamics of ether. We know how volatiles work. I’m not going to labour the point. Gavit A receptors messing around. Cardiovascular side effects of ether. Ether was a positive chronotrope.
Pharmacodynamics of Ether
09:20-14:20
Summary:
• CVS: Positive chronotrope, vagolytic, SNS stimulation
• Respiratory: Increases rate, decreases depth, bronchodilator but irritant
• CNS: Dose-dependent ICP increase, 50% nausea/vomiting, has analgesic effect
• Metabolism: 2-3% hepatic (to acetaldehyde, alcohol); 97% lung excretion
• Wide safety margin due to high blood-gas solubility (slow onset/offset)
And at concentrations between 2% and 82%, ether will go boom, kaboom, boom. Much like those penguins from Madagascar who like to blow stuff up. Ether, maybe they could have used it. So pharmacodynamics of ether. We know how volatiles work. I’m not going to labour the point. Gavit A receptors messing around. Cardiovascular side effects of ether. Ether was a positive chronotrope. by being vagalytic and stimulating the sympathetic nervous system. You can imagine that back in the day when you’re doing slightly hairy anesthesia and you don’t really have a clue what you’re doing, it’s actually probably quite good that it jazzed up your cardiovascular system a bit. It probably is better than the opposite where someone looks like they’ve died because you wouldn’t be very good at telling if they’d died. From an inotropy perspective, in vitro it seems to be a negative inotrope, but in reality, i.e. in vivo, the SNS activation has a positive inotropic effect. It doesn’t increase your odds of arrhythmias. It vasodilates your coronaries and you might think it’s going to vasodilate yourself entirely. It does but the sympathetic nervous system activation which occurs in deeper planes of anaesthesia pushes your blood pressure back up again. So quite a forgiving thing to use when you don’t really know what you’re doing. So in light planes it drops your blood pressure but your patient is going to be wriggly. When you get them accidentally too deep you actually see it increase and heart rate. Which, you know, nice. It doesn’t sensitise the body to catecholamines. From a respiratory side effect profile, it, like most other volatiles, increases the rate of breathing but decreases the depth of breathing. Eventually we will talk about those respiratory nuclei in your brainstem and the pneumotoxic centre, blah blah blah. Not today. That’s when we get onto physiology in three and a half decades time. And the parenchymal effects, it’s again going to inhibit hypoxic is a bronchodilator, but it is quite irritant and patients are really prone to coughing and breath holding, especially if you ramp up the dose too quickly. So if you accidentally slosh a bit too much ether on that rag or into that teapot, then they’ll hack and cough and breath hold and then you’ll find yourself in a tricky plane of anaesthesia. CNS effects, as you would expect, it has a dose dependent effect on your intracranial pressure via causing cerebral basodilation. It caused nausea and vomiting in 50% of the patients that were exposed to it, which is really quite high. But interestingly, ether has an analgesic effect. So it’s not just an anaesthetic puncture on the end, but as Dr Crawford long noticed, people were, you know, falling off ladders, having smashed some ether and not noticing. And it also causes hypersalivation. And this is probably why there was the hysteria historic use of atropine as a pre-med that crept on for quite some time. From a metabolic perspective, ether stimulates gluconeogenesis, i.e. making your glucose in your liver, and may also cause a type B lactic acidosis state. But I don’t know if Dr. Long was checking folks lactates. It causes renal artery vasoconstriction, potentiates neuromuscular blocking agents, again causes uterine hypotonia, and some of the literature I’ve read, confirms that ether had a wide safety margin and this was in part because of the side effect profile from cardiovascular angle but also the fact that it takes quite a long time to get ether into the person because of that really really high blood gas solubility so the onset time is a really really really hecking long which means that it’s kind of hard to overshoot because any swings in dose are buffered by that large blood gas solubility so Absorption. Now, this is quite funny, because I’ve made jokes about absorption and volatiles before, but actually folks used to drink ether, and it had effects. Particularly, I’m sure it cured stomach ghosts, but also made people feel a bit drunk. So it undergoes, in the liver, 2-3% metabolism into acetyl aldehyde, alcohol, acetic acid, and CO2. This is probably why drinking ether, and therefore having quite a significant first-pass metabolism, would make people feel a bit boozed up because it’s being converted to alcohol. And therefore by virtue of the fact that 2-3% undergoes liver metabolism, 97% of ether is excreted by the lungs. Although remember folks that high blood gas solubility coefficient means that it’s going to take quite some time to get out of people. Remember 12 versus the 0.45 of desflurane. So that was a fun jolly with ether and we’ll come back to ether in the latter half of this episode when we’re going to get out of the way. We talk explosions, which is what we’re all here for. But we’re just going to quickly dab onto n-flurane. And I can tell you, likely the most important thing you need to know about n-flurane for the FRCA primary exam is that it is a structural isomer of isoflurane. Now, it is a clear and colorless non-flammable liquid. Its MAC is 1.6%. Its cardiovascular effects include a tachycardia, negative ionotropy. It sensitizes the myocardium to
Enflurane
14:20-15:49
Summary:
• Structural isomer of isoflurane (key exam point)
• MAC: 1.6%; Non-flammable
• CVS: Tachycardia, negative inotropy, myocardial sensitisation to catecholamines
• Respiratory: Bronchodilation but significant respiratory depression
• CNS: Can induce seizures, increases ICP and CBF
• Metabolism: 2.4% hepatic, 2.4% urine/sweat/faeces, rest via lungs
We talk explosions, which is what we’re all here for. But we’re just going to quickly dab onto n-flurane. And I can tell you, likely the most important thing you need to know about n-flurane for the FRCA primary exam is that it is a structural isomer of isoflurane. Now, it is a clear and colorless non-flammable liquid. Its MAC is 1.6%. Its cardiovascular effects include a tachycardia, negative ionotropy. It sensitizes the myocardium to and coronary and general artery vasodilation. It has those classic respiratory side effects of being bronchodilating but it causes quite significant respiratory depression compared to the increased rate, reduced depth breathing that you might see with other volatiles. CNS wise, nflurane also can induce seizures and increases intracranial pressure and cerebral blood flow in a dose dependent manner. like other volatile anaesthetic agents. It is 2.4% metabolized in the liver, a further 2.4% is eliminated via the urine, sweat and feces and the rest is cleared via your lungs. Anyhow, time for a brief mention about the sponsors of Gas Gas Gas, Teach Me Anaesthetics. These guys are behind an excellent single best answer question resource. Now firstly, I took the joyful, challenging and intermittent bashing, through the 1100+ questions they have written for the FRCA primary exam. Now I secretly did love it and there are plenty of questions to test your knowledge. Now when I was studying for the primary it didn’t exist but there were other packages online. These cost a lot more and they didn’t really seem to reflect the content of the exams and whilst doing these I came across rehashed MRCP questions and I wasn’t terribly inspired and really I was a bit cheesed off. Whereas these single best answer questions have been built from the ground up and they have not sniffed an MCQ in a
Fire Triangle and Key Definitions
23:00-28:14
Summary:
• Fire requires: Heat source + Fuel source + Oxidiser (O₂, N₂O, NO, H₂O₂)
• Ignition: Initiation of combustion requiring activation energy
• Combustion: Exothermic chemical reaction producing heat and light
• Flash point: Minimum temperature for ignition with external flame
• Auto-ignition temperature: Temperature for spontaneous ignition
• Deflagration: Subsonic combustion with propagating flame front
• Detonation: Supersonic combustion with compressive shock wave
• Explosion: Rapid volume expansion of gas in short timeframe
First Prof of Anesthesia in the United Kingdom back in the 1940s. And he identified that there was a bit of a gap in the knowledge of the anaesthetists of the time. Very practical folks. But physics, it was sort of pulled together from archaic physics and chemistry profs, as opposed to something that’s a bit more focused for the anaesthetists. You know, the jobbing anaesthetist who needs practical ideas as opposed to slightly nebulous ideas. And so physics for the anaesthetists, including a section on explosions, was born. It’s gone through a number of permeability. It is still in copyright and I would thoroughly recommend it and there’s a link to the Welcome Library page for this book in the show notes. So Robert MacIntosh, developer of the Mac Blade amongst other things. So definitions or death, there’s quite a few new words that we’re going to bring about in this episode and just some commonly used terms that we really want to nail down. So a flame. What’s a flame? A flame is a hot glowing body of ignited gas that is generated by something These combustions produce light. This light is the flame you see. What is a cold flame reaction? This is an oxidation combustion reaction of a mixture that will only sustain itself whilst there is a source of heat supplied. So as soon as you remove that lighter, it stops burning. Now there’s a deflagration and conflagration. A conflagration is an extensive fire which is sort of wrecking the place. You know, a hillside alight in a white You might be able to argue that if there was a whole ward on fire, that ward was in a conflagration. Whereas a deflagration is one of these oxidation-combustion reactions that is burning with a sudden flame, it’s rapid, and is generally in a controlled system or in a contained space. I’ve said combustion a few times there. What is combustion? Combustion is a rapid exothermic oxidation reaction between a fuel and an oxidant. Oxidant usually being oxygen. That releases significant energy as heat and light. Often producing oxides, carbon dioxide and water as products of that combustive process. Flashpoint. This is something that is worth a definition but it’s not going to come up again in the episode. A flashpoint is a property of a liquid fuel whereby you draw a flame across the top of it and see if you ignite the volatile elements of that compound off gassing or vapourizing from that liquid. If you have quite a cold volatile compound, you’re going to get less vapourisation on its surface. You might not see ignition, whereas a nice warm volatile compound more likely to ignite. The flash point is that point where it goes from being ignited with that flame drawn across the top of it to not being ignited. And then what is an explosion? So an explosion is really anything that achieves a volume of expanding gas in a very short time frame. The shorter the time frame, the bigger the So mechanical, this is overloading something with pressure. So for example, the baby milk bottle analogy in the microwave that was an excellent accident on my part, but also if you were to close the safety release valves on a vacuum insulated evapourator in a hospital and heated it up, eventually it would go bang. Remember that one litre of liquid oxygen would much prefer to be 861 litres of gaseous oxygen when it’s There are chemical explosions. When we say chemical, these are TNT, C4 type stuff, high explosives. And then deflagrating chemical explosions, i.e. low explosives. That’s what tends to happen in anaesthetic land. And then nuclear explosions. Rapid release of huge quantities of energy, causing heat expansion of the gas around it. And that’s due to that chain reaction of neutrino badness. Go watch Oppenheimer if you’ve not seen it. If you’ve seen it, you should go watch it. If you’ve seen it, watch it again. It’s great. It’s on iPlayer at the moment. Make sure to turn the volume up. And then detonations. So detonation is combustion of a substance, which is a sudden and extremely rapid propagation of the combustion that’s occurring, giving rise to a shock wave. And what’s a shock wave, you ask? A shock wave is a sharp change of pressure in a narrow region traveling through a medium. We’ll presume air. for this example, caused by an explosion or by a body moving through that medium faster than sound travels through that medium. Hence, you get a sonic boom of a jet aircraft above Mach 1 howling through the atmosphere. Great, so those are our definitions. Hopefully this will smooth out the rest of the episode. So, we’re going to talk fire first, then we’re going to talk explosions, because naturally they do lead on. So, for a fire to get 11 A-stars at GCSE, It requires the correct combination of three things. A heat source, a fuel source, and an oxidiser source. Classically oxygen, but it can also be nitrous oxide, nitric oxide, and hydrogen peroxide. Noting that all those three molecules contain oxygen. Heat sources, I’m going to go into more detail, but just think a spark, a flame, or sufficient compression of a gas. If you compress something, you will make it hotter.
Fire Safety and Mitigation in Anaesthesia
28:14-32:12
Summary:
• Patient factors: Minimise O₂ concentration, avoid leaks, careful with nasal specs
• Remove N₂O from theatre (disconnect and discard)
• Surgical prep: Ensure alcohol solutions dried, no pooling
• Laser safety protocols essential
• Equipment: Tested, earthed, no tingling sensations
• NEVER grease Bodok seals – can ignite under pressure with O₂
• Static mitigation: Conductive floors, antistatic shoes, humidity control
It requires the correct combination of three things. A heat source, a fuel source, and an oxidiser source. Classically oxygen, but it can also be nitrous oxide, nitric oxide, and hydrogen peroxide. Noting that all those three molecules contain oxygen. Heat sources, I’m going to go into more detail, but just think a spark, a flame, or sufficient compression of a gas. If you compress something, you will make it hotter. That’s why your bicycle pump gets warm. If you’re very lazy and you try and pump up a flat car tyre at home instead of taking it to the garage with a bike pump, taking ages, your bike pump gets quite warm. Other perhaps slightly more common sources in an NHS hospital would be electrocortery, diathema lasers, magnifying glasses maybe, or optics. And in true NHS fashion, everyone knows of a slightly dodgy electrical socket with some tape over it saying do not use. You shouldn’t use that. And nor, arguably, should you be toasting bread using an open flame or modern toasting devices. These are indeed notoriously sparky and will be sufficient for triggering deflagrations and explosions and fuel sources. So we know that we shouldn’t pile up notes outside fire exits in hospitals, but in theatre. Bowel gas can go up in smoke. The surgical prep. Drapes that we use can melt, burn and cook. And obviously, if we’re anesthetizing people with ether, cyclopropane, divinyl ether, then we might see one or two ignitions of that. So how do we mitigate these problems? Because in the exam, they’re asking you an avivor about fire. They want you to probably quote those three, the triad of fire that you need, you know, the heat, oxidizers, and fuel. And then what do you do to reduce the risk? So it depends situationally on what’s going on. and you could probably break it down into patient environment and surgical. Patient being, be as cheap as the oxygen as you can, don’t leak loads of oxygen out around a face mask. If you were in recess, don’t leave the 15 litres blowing under the covers. And on the wards, nasal specs under the sheets and patients deciding that they are going to have that damn cigarette. Doesn’t end well. Unplug the nitrous oxide from the anaesthetic machine, throw away the pipes, then no one can use that and cause bother and the polar bears will thank us. Keep an eye on the surgeons and sloshing around that alcohol solution, making sure it’s dry and cleared and there’s nothing pooled down the sides of the patient. Very significant application of fire safety when utilising lasers in theatre and being cautious about setting fire to the patient’s body hair. Surgical equipment itself should be safe, tested and not causing any tingling when you touch it, which would suggest earth leakage. And from an anaesthetic equipment perspective, do not grease your bodoc seals as much as you are tempted to. get that can of grease which is obviously in every anaesthetic room for any emergency lubrication problems but if you plonk grease on your neoprene with metallic ring bodox seal which goes between your oxygen tank and the yoke on your anaesthetic machine the compression effect of the tank pressure when opened can trigger fire around your bodox seal which has lots of oxygen being supplied to it as well however cool having flames howling out of the back of your anaesthetic machine might sound. There’s a lot of other stuff dangling around the back of an anaesthetic machine that also probably has a propensity to get burny in oxygen. So don’t grease your bodox seals folk. Now there’s a great BJA article on fire and managing fire which is linked to in the show notes. So we’ve talked about mitigating fire but what about actually managing a fire? So this depends on whether or not the patient is on fire, a staff member is on fire, the room is on fire, or There is an airway fire. We’re going to focus on airway fires because, you know, theatre sisters should be able to put out the scrub nurse, hopefully, whilst we manage that anaesthetic. So with an airway fire, sometimes it’s quite challenging to spot it. There might be a flash, a pop as your endotracheal cuff goes pop whilst it expands in the heat, a funky smell, or you might see some smoke. Once you’ve identified a fire, the second step is declaring or hollering fire.
Managing an Airway Fire
32:12-35:11
Summary:
• Recognition: Flash, pop (cuff), funky smell, smoke
• Immediate actions:
1. DECLARE FIRE – shout “Fire!”
2. Stop ventilation
3. Deflate and remove airway devices
4. Slosh copious saline into airway
5. Surgeon removes debris
6. Resume ventilation with LOW FiO₂ (bag-valve-mask preferred)
• If not contained: CO₂ extinguisher, fire alarm
• Post-fire: Inspect airway (bronchoscopy/laryngoscopy), airway toilet
• Re-intubate and keep sedated if airway burns suspected
• Leak test before extubation (tracheal swelling risk)
There is an airway fire. We’re going to focus on airway fires because, you know, theatre sisters should be able to put out the scrub nurse, hopefully, whilst we manage that anaesthetic. So with an airway fire, sometimes it’s quite challenging to spot it. There might be a flash, a pop as your endotracheal cuff goes pop whilst it expands in the heat, a funky smell, or you might see some smoke. Once you’ve identified a fire, the second step is declaring or hollering fire. And then in a coordinated manner, a number of things have to happen. You should stop ventilating the patient. Deflate and remove airway devices. Slosh copious amounts of saline into the airway. When there are operations involving airway fire risk, i.e. ENT lasering, a jug of water must be available on the scrub trolley for such a thing. The surgeon should remove any remaining debris once saline is in, i.e. plastic that’s cooled down, a fragment of etc. You should resume ventilating the patient with low oxygen fractions in case there’s anything smouldering inside the patient, ideally with a bag valve mask to avoid an excessive oxygen. Naturally, if you’re maintaining anaesthesia with volatile, then you will have to use your anaesthetic circuit, but probably worth dialing down the oxygen, rolling that flow rate right high, and trying to flush the oxygen out of your circuit. And at that point, you probably need to make some decisions about next steps. If the fire is not contained, and is spreading over the patient, then a CO2 fire extinguisher would certainly be a worthwhile thing to blast all over the patient, try and put them out, and maybe trigger a fire alarm. If you’ve been successful in attaining control, then you need to inspect the airway for injuries, and that could either be an aesthetist-led bronchoscopy or surgeon-led laryngoscopy, although arguably you need to have a look further in if you think there has been a fire down the patient’s airways, because There might be distal inflammation and redness which we need to think about. It’s certainly worth conducting some airway toilet to make sure that no debris is lurking down there, re-intubating the patient or intubating the patient if they weren’t tubed to begin with, and keeping them sedated if there’s any whiff of a suspicion of airway burns as they will end up with gas exchange complications and ARDS. And if their trachea starts swelling up, you’ll just make your life harder to intubate them. When you’re going to extubate them, remember to do a leak test to actually make sure that that trachea isn’t just incredibly sweltered and pressed on your tube. Cool. Right. So we talked about airway fires and we talked about mitigating airway fires. But what about more broadly how fuel mixes may become ignited, i.e. sources of ignition? So there’s auto-oxidation or auto-oxidation. And this actually happens with ether. Auto-self-oxidation. Well, you might have guessed oxidation means oxidation. But once upon a time, ether, pure, in a glass bottle, left on a shelf in the sunlight, would produce some hydrogen peroxides, either through impurities or the nature of the molecule itself, which are really labile and act like detonators. That meant that when you picked up and accidentally shook a bottle of ether that you’d left on the shelf, it could just go kaboom. Ether that was supplied to hospitals and used for anaesthesia was kept in containers that
Auto-oxidation and Sources of Ignition
35:11-39:11
Summary:
• Auto-oxidation: Ether in sunlight produces hydrogen peroxides (detonators)
• Historical: Ether stored in light-protective containers with antioxidants
• Auto-ignition temperatures: Wood 300 °C, Isopropyl alcohol 399 °C
• Hospital ignition sources: Diathermy, lasers, faulty electrics, static discharge
• Static prevention: Conductive materials in masks, humidity maintenance
• Historical: Theatre humidity and conductive flooring critical for ether safety
But once upon a time, ether, pure, in a glass bottle, left on a shelf in the sunlight, would produce some hydrogen peroxides, either through impurities or the nature of the molecule itself, which are really labile and act like detonators. That meant that when you picked up and accidentally shook a bottle of ether that you’d left on the shelf, it could just go kaboom. Ether that was supplied to hospitals and used for anaesthesia was kept in containers that light exposure with antioxidants as part of that liquid, therefore making it all rather a bit more safe. Spontaneous ignition. Well, it’s not quite spontaneous ignition. If you have a gas and you put it in a box and then you just heat that box up, eventually you will warm that gas through to a sufficient point whereby it will spontaneously start to combust. And when I say gas, I mean fuel mix, because if you just had pure butane in a box and heated it and heated it and heated it, but there’s no oxidizer, then you can You would get no combustion, wouldn’t you? Some very few gases actually spontaneously ignite at room temperature. These are quite niche things. Most stuff generally needs to be rather warm to ignite. And you can see why this is logically the case, because otherwise life on this planet wouldn’t be as we know it if trees just spontaneously erupted. So wood will auto-ignite at 300 °C. And isopropyl alcohol, that’s stuff in chlorhex wipes, at 399 °C. So whilst I appreciate that NH hospitals sometimes feel like they’re at 399 °C, the fact that the claw hex isn’t going up in smoke suggests otherwise. And then obviously in hospital sources of ignition, we’ve covered them before open flames. Very few theatres have open flames in them. Although once upon a time they did used to have fireplaces to keep the theatre warm. You could do your toast on it, but if you were to spill some ether near the fireplace, Bad News. Cigarettes, obviously, were a bit of a concern once upon a time. Nowadays, laparoscopes, diathermy, and there’s always going to be risks of arcs from faulty switches. And we all have to walk around, or at least on paper have to walk around, in shoes that prevent static buildup. We have floors that are conductive that dissipate static, because a static charge built up in one object that’s transferred to another object can cause a spark and cause ignition. Now it’s only non-conductive, poorly earthed objects that will do this, because if you’ve got a metal bar and you somehow induce a static charge in it, but that static charge is connected to you by your hand, you will equalise in charge with that bar, and therefore you won’t get a discharge between them. Whereas if you’ve got a rubber balloon and you’ve rubbed it and rubbed it and rubbed it and rubbed it, it will hold a charge because it’s quite a good insulator. So the face masks of old, the big rubber face masks of old, the material in that was altered to make them more conductive, i.e. less effective insulators. And this is why also humidity in theatres was a thing. You would maintain sufficient humidity in your theatre to keep a very fine layer of moistness around the place because that fine layer of moistness improved conductivity and therefore reduced static build-up chances. All very interesting. That’s why we used to be obsessed with having Now, not really an issue is it? We don’t use things that explode. Makes our lives safer and naturally less exciting. So, explosives in anaesthesia. The explosions that were seen in anaesthesia were not the absolute fastest, i.e. like dynamite or TNT. Generally speaking, it’s a deflagration that gets carried away and detonates because it is operating in the conditions that would enable it to detonate. And we’re going to go through what a combustion reaction looks like on paper. We’re going to talk about things that influence reaction rates and talk about what it means to have a detonation within a breathing circuit. And then that’s the episode over. And you’ll just have to wait another two weeks-ish. I do have a newborn, but you know, we’re going to try our best. So, the explosives. Once upon a time in anaesthesia, we had ether, which is called diethyl ether, remember? Then we had divinyl ether, which unfortunately is,
Explosive Agents in Anaesthesia
39:11-42:11
Summary:
• Diethyl ether: Explosive in O₂ 3-33%, burns in air 1.83-48%
• Divinyl ether: More explosive than diethyl ether
• Ethyl chloride: Explosive 4-67% with O₂, 3.8-15% in air
• Cyclopropane: Highly flammable (historical use)
• Nitrous oxide: Supports combustion, can burn hotter than O₂ alone
And we’re going to go through what a combustion reaction looks like on paper. We’re going to talk about things that influence reaction rates and talk about what it means to have a detonation within a breathing circuit. And then that’s the episode over. And you’ll just have to wait another two weeks-ish. I do have a newborn, but you know, we’re going to try our best. So, the explosives. Once upon a time in anaesthesia, we had ether, which is called diethyl ether, remember? Then we had divinyl ether, which unfortunately is, more explosive again. Ether in itself will not explode when mixed with air only, which is the case for most things in anesthesia. But ether in an oxygen mixture, obviously you may have a mix of oxygen, air, or I should really say oxygen, nitrogen, plus ether. And if you have more than 21% oxygen, you could say that as an oxygen-rich mixture. But in a range of between 3% and 33% ether in oxygen, you will have an explosive that will happily detonate. Whereas ether in just air will burn from a range of 1.83 up to 48% ether. Burn or nice word here is deflagrate. Other things that go boom. Ethyl chloride we mentioned. That’s less available these days but I’m sure you might have seen those glass bottles with a little spray lever around that you can use to test cold and chill skin to put a cannula in. In the presence of oxygen ethyl chloride will form an explosive mixture with A mere 4% ethyl chloride up to 67% ethyl chloride. Ehyl chloride mixed in air can also explode though 3.8 to 15% noting you can’t if you had 67% ethyl chloride it would be crowding out your oxygen as well as your nitrogen therefore the availability of the reactants will be less. Cyclopropane now not in use but previously was and that is highly flammable and a oxidizer that we still can get our hands on is nitrous oxide. This supports combustion and can burn hotter than a solitary oxygen mix purely because it’s more oxygen available. As you heat it up, the oxygen is liberated. Obviously, it does leave nitrogen behind. And this is why nitrous oxide, especially Need for Speed Underground 2 and other such games, is used to make you go briefly faster with your car. Zoom zoom. Okay, so if you were to try and write down a combustion reaction on a bit of paper to kind of break it all down and draw some logic in an exam. You would write gases A and B, those are your reactants, plus heat added leads to a chemical rearrangement producing products D and C, plus minus heat plus minus photons. When we say heat, that’s heat energy, but you could also make an argument that you’re actually emitting electromagnetic spectrum of which photons are one, but infrared radiation is another. This probably hopefully harks back to A-level GCSE chemistry where we talk about reactants and products etc. Just remember that you’re adding heat to alter bonds because you need to achieve a sufficient activation energy of a reaction to trigger its chemical rearrangement. This chemical rearrangement classically steps down from a set of more reactive molecules to a set of less reactive more stable molecules. So a good example that perhaps elaborates on activation energy.
Combustion as a Chemical Reaction
42:11-47:00
Summary:
• General equation: A + B + heat → C + D ± heat ± photons
• Activation energy required to break/rearrange chemical bonds
• Wood combustion example: Different components burn at different temperatures
• Incomplete combustion: Creates smoke, creosote buildup (chimney fire risk)
• Stoichiometric mix: Ideal fuel-to-oxidiser ratio for complete combustion
• Rich mix: Excess fuel (incomplete combustion)
• Lean mix: Excess oxidiser (incomplete combustion)
This probably hopefully harks back to A-level GCSE chemistry where we talk about reactants and products etc. Just remember that you’re adding heat to alter bonds because you need to achieve a sufficient activation energy of a reaction to trigger its chemical rearrangement. This chemical rearrangement classically steps down from a set of more reactive molecules to a set of less reactive more stable molecules. So a good example that perhaps elaborates on activation energy. is burning wood. So there are multiple flammable things in a lump of wood. It isn’t just that block of carbon that we all imagine, but it’s got sugars in it, it’s sap in it, and it’s got things like creosote in it. And these all burn at different temperatures, i.e. they have different activation energies. We’ve all witnessed this when we’ve started a fire, and it’s all smoky and crap and everyone’s spluttering. And once you get it roaring, all that smoke dissipates. This is because you are successfully achieving near maximal combustion of all the molecules. So if we take that step further and now we’re thinking about our wood-burning stove, it would be inefficient to have lugged all that wood home, stacked it, dried it, put it in your wood-burning stove and then just sit there and watch it smoke like shit because you’re wasting half of the energy. You need to burn all of that material. A, if you don’t, you’re wasting heat. B, that cold, grotty smoke. will rise through your chimney or flue and cool down. And if it cools down, it will stick to your chimney. And you get creosote buildup. That’s why chimney sweeps exist. That creosote buildup, when you finally get a roaring fire going, will trigger a flue fire and you set your chimney on fire. And that’s bad news. Therefore, remember, you need sufficient activation energy for all the steps and all the things being ignited in your reaction. Otherwise, you’ll have partial burning and loads of Spare bits of combustion knocking around that could be used. So now we’re progressively thinking more about combustion as reaction rates. There are a number of things that influence reaction rates in a system. Now, I don’t know of any enzymes that get involved in burning, but I can tell you that the warmer your system is, the faster it’s going to work. The nature of your fuel mix has a significant influence on reaction rate, and the physical environment within which your combustion reaction is taking place is important for two reasons. An enclosed physical environment with burning that’s heating the gases in the environment will cause compression and actively warm things further by increasing pressure but also trapping heat inside therefore enabling the reaction to continue trundling forward getting hotter and hotter and hotter. We’re going to talk about fuel mix first. You would have a quantity of reactants, your wood and your oxygen, and a quantity of products. And the ratio of wood to oxygen is completely on the dot that everything gets burned and consumed. That’s a stoichiometric mixture, i.e. one that is going to be completely consumed in the reaction. Stoichiometric mixtures burn the best and the fastest. If you are swinging to either direction, On the sides of that stoichiometric mixture, you have a richer mixture if you’ve got too much fuel, not enough oxygen, and a leaner mixture if you’ve got too little fuel and the same amount of oxygen. You might have come across rich and lean fuel mixes being talked about if anyone’s explained how a carburettor works in a motorbike, for example, whereby you can slightly alter a pin and change the nature of the fuel mix. If your motorbike’s kind of spluttering and struggling to get going and it’s actually running a bit lean, adding a little bit more fuel might make it idle smoother, but you consume more fuel that way. If you have a really rich mixture howling through your carburettor into your engine, you will get incomplete combustion and lead to debris and furrowing up and all sorts of bother within your engine. So what about heat gain and heat loss and how that contributes to a reaction? Only 10 °C doubles your reaction rate, which blows my mind actually. If your losses in your fire are too high, then you have an imbalance that leads to failure of propagation of that fire, or within the system I’m going to explain in a minute, a failure of that burning wave front to progress through your combustible gases. If you have non-combustible materials within your system that soak up heat, that’s going to slow you down.
Deflagration vs Detonation
47:00-52:00
Summary:
• Deflagration: Subsonic flame propagation, heat transfer ahead of flame
• Detonation: Supersonic propagation, compressive shock wave heats gases
• Tube dynamics: Closed-end tubes increase pressure, accelerate flame
• Vertical vs horizontal: Flame rises faster (lighting from bottom accelerates)
• Transition to detonation: Requires optimal fuel mix, insulation, tube length
Tons of Nitrogen knocking around. Nitrogen doesn’t get involved in the burning at all, but it still has to be warmed up before that reaction can get truly going. Is there a route for heat to be conducted away from that burning system? Now, that might be a goal if you’re burning gas in a boiler to warm water, but if you’re burning gas to speed things up, it’s not so helpful. And then reaction vessel itself, so I spoke about that briefly earlier. If you have a a combustive process occurring within an enclosed space. It’s going to get hotter and hotter and hotter and go faster and faster as long as fuel and oxidizer remains present. This is why fire services are very hesitant about opening doors. If you’ve got a fire behind a door, that room is probably rip-roaringly hot but has consumed most of its oxygen but it may have loads of fuel spare. You open that door, you’re going to absolutely immediately suck oxygen into that room. and you’re going to get engulfed in flames piling back out of that room. I’ve got an example of that at the end of the show just to close on. Not quite me being burned alive by a fire but almost no. So us as anesthetists we’re less interested in how a fire might behave in a bedroom but we are more interested in how a fire, a deflagration or an explosion might behave within our ventilatory apparatus. Our tubes, our pumps, our anaesthetic machines. So first off, we need to put our imagination hats on. We’re going to imagine a tube, and it’s open-ended, both ends. We’ve got an A end and a B end, and within that tube contains a flammable gas mixture. We’ve tipped some ether in there. We’ve sloshed it up and down, and it’s evapourated off and filled that tube with a bit of ether, but there’s still some air in there as well. And then we light point A. Four things may occur in our tube system here. The flame sputters, and it fails. There is insufficient heat to maintain combustion or the fuel mixture is non-conducive to combustion. It may simply ignite the gas on point A and you’ll see a wave front of flame transit across your tube to point B. The ignition may near immediately cause a burst of flame and heat from either end of the tube almost imperceptibly fast. That’s tipping towards explosion and detonation. And the fourth option would be the mix fails to ignite at all. And that could be because there’s simply too much ether and not enough oxygen. So as long as that mixture can self-maintain its combustion with the heat it generates, you will end up with a flame journeying across from A to B. So that could be an ignition from your anaesthetic machine journeying through your circle circuit to the patient end. Now the nature of the tube does influence the nature of the combusting wave front. through it. For example, I’ve described a tube which has two open ends. But if you have a closed end and cause an ignition at that closed end, you will end up with greater rates of heating and expansion of that wavefront. There’s no blowback, increasing pressure behind it, and a more rapid transit of flame. That’s why a rifle shooting a bullet isn’t open-ended at the back. It’s a closed barrel. with one end that opens in order to allow all that force to drive that round out of the end of your rifle. So let’s imagine our tube again. We can alter the properties of that system to either support combustion or inhibit combustion. And we’re interested in explosions here, so we’re going to try and support combustion. And by that virtue, we need a mixture that’s nearer stoichiometric than not, i.e. not rich or lean. There aren’t other materials and gases in there uninvolved in that reaction. They’re just going to waste energy, i.e. nitrogen floating around in there. We might want to insulate that tube to help contain the heat that is generated. Eventually, we will find ourselves in a situation where we alter our propagating flame, our deflagration down that tube, into an explosion down that tube. That transition from deflagration to explosion, involves detonation and that’s when a deflagration accelerates to the point where it tips into generating a compressive shockwave that heats the gases ahead of the wave front therefore facilitating faster and faster and faster combustion. Long tubes more likely to achieve that shift from deflagration to explosion as long as the fuel mix is sufficient and whichever angle the tube is that also is important. So if you have a vertical tube as opposed to a horizontal tube and you light the top and you expect that flame to track downwards, so I can imagine, and it definitely bears out in reality, it’s quite slow. If you were to light that tube from the bottom, it’ll go all the way up that tube very, very fast, and you’re more likely to tip into a detonation from a deflagration process. Why are we chatting about detonations in tubes and deflagrations?
Detonation in Anaesthetic Circuits
52:00-53:34
Summary:
• Detonation pressure: 25-30 atmospheres = 3,000 kPa = 30,000 cmH₂O
• Compare to normal ventilation: 25-30 cmH₂O peak pressure
• Effect: Blast lung injury (similar to explosion/grenade victims)
• Circuit damage, patient lung injury beyond simple airway burns
is that also is important. So if you have a vertical tube as opposed to a horizontal tube and you light the top and you expect that flame to track downwards, so I can imagine, and it definitely bears out in reality, it’s quite slow. If you were to light that tube from the bottom, it’ll go all the way up that tube very, very fast, and you’re more likely to tip into a detonation from a deflagration process. Why are we chatting about detonations in tubes and deflagrations? in tubes. Well, that’s because when a patient’s attached to that tube, it’s arguably quite bad news. Beyond a patient simply receiving airway burns, if there’s a detonation inside your anaesthetic circuit, obviously it’s bad for your anaesthetic machine, it’s bad for the patient’s lungs because they are exposed to a pressure wave. When you achieve a detonation, it will deliver a pressure of 25 to 30 atmospheres, which is quite a lot. Remembering that one atmosphere of pressure is a 30 atmospheres of pressure therefore would be 3,000 kilopascals of pressure which is the equivalent of about 30,000 centimeters of water of pressure and when we start having kittens about plateau pressure or a peak pressure of 25 to 30 centimeters of water you can see that that’s quite a lot of pressure to expose to someone’s lung and they will get blast lung which is something you can recognize in people are victims of gas explosions or grenades in enclosed spaces in conflict. In this episode, so far, we have spoken about ether and fluorine, fire, what causes fire, mitigation of fire, airway fire, talked about combustion as a chemical reaction, used wood as an example, spoke about auto-ignition, auto-oxidation, and touched on detonations in,
Personal Anecdote: Butane Bottle Incident
53:34-56:24
Summary:
• Age 16: Filled 2L Coke bottle with butane from lighters
• Initially: Rich mixture (excess fuel) burned slowly at tip
• Accidentally aspirated air while reigniting
• Created stoichiometric butane-air mixture
• Result: Rapid deflagration/detonation, hand blisters, melted bottle
• Lesson: Demonstrates importance of fuel-oxidiser ratio
In this episode, so far, we have spoken about ether and fluorine, fire, what causes fire, mitigation of fire, airway fire, talked about combustion as a chemical reaction, used wood as an example, spoke about auto-ignition, auto-oxidation, and touched on detonations in, Anaesthetic Circuits. The last thing I want to do is tell you a story about how, as a young idiot, I caused what now probably sounds a little bit like a detonation to me. So I took a two-litre Coca-Cola bottle and obviously drank the Coca-Cola, because who doesn’t need all that sugar when they’re 16? And then I filled that Coca-Cola bottle with butane from two lighters, I think it was. I then lit that, and it burned slowly with a flame from the end, a bit like a a butane molotov cocktail. I’m sure quite a few people have also done this. And if you squeezed the bottle, it would eject some of the butane and you would get a pretty good arc of flame come out of it, which was great fun. But intermittently it would go out. So as I was fiddling around trying to reignite the mix, I squeezed the bottle, aspirated a load of air inside it alongside the flame. This achieved quite a good mixture of flame, butane, air, And it all ignited as opposed to the air butane interface at the tip of the bottle that was causing my little pilot flame sort of effect that I was seeing initially. This ejected from the bottle all over my hand at a blistering rate with a bit of noise as well if I remember correctly. This was quite a few years ago now. And A, it melted the Coke bottle a little bit and B, it blistered the crap out of my igniting hand. So naturally, if you would own up to this as a child, my hand became better in a week or so. But I think this demonstrates that initially, the mixture in that Coca-Cola bottle was really rich. So it wasn’t actually conducive to burning. It didn’t have enough oxidizer in it. It didn’t have enough oxygen. But eventually, by mucking around and running around and spraying flames at my mates, I had a sweet spot where I inadvertently probably created the stoichiometric butane air mix. And it went vwomp. burning in a much rapid and hotter manner than it had initially been doing so. So after this, I was far more careful and ensured to point the bottle at my friends and not myself. I was a young idiot and I may still be an idiot now seeing as I managed to blow the door open on the microwave a number of days ago. But that was the gas, gas, gas, slightly protracted Christmas special. Why wouldn’t you want to think about ether and nflurane and explosions at Christmas? Christmas instead of having to stress completely about the FRCA. Some of this stuff is certainly FRCA relevant and a number of it is just purely for interest and curiosity. I particularly liked the teapot ether action in Clifton in Bristol. That just makes me chuckle. British people are like, well, what else can we use a teapot for? As I know, ether. Love it. Check out the show notes. If you do stumble across physics for the anaesthetist in a charity shop near you, treat yourself.
Closing Remarks
56:24-58:00
Summary:
• Episode balances FRCA relevance with curiosity
• Check show notes for references and diagrams
• Support options available on website
• Reminder: Study steadily, don’t overcook revision
Christmas instead of having to stress completely about the FRCA. Some of this stuff is certainly FRCA relevant and a number of it is just purely for interest and curiosity. I particularly liked the teapot ether action in Clifton in Bristol. That just makes me chuckle. British people are like, well, what else can we use a teapot for? As I know, ether. Love it. Check out the show notes. If you do stumble across physics for the anaesthetist in a charity shop near you, treat yourself. Why not? It’s a lovely book to have on your shelf. So I hope you’ve all had a lovely Christmas. You’ve had an opportunity to celebrate some time and take some time with your families and enjoy some relaxation in this holiday season. Happy New Year. I’ll see you in the hospital soon enough. I’ve got to try and survive pat leave first. Ahoy, Team Anesthesia! You’ve survived yet another episode of Gas, Gas, Gas. Now, if you found it useful or harrowingly awful, Please leave a comment or ping off an email if you think I need to square something away. Now there are a bunch of ways to support the costs of GasGasGas. From buying me a coffee, to venturing forth via an affiliate link, to the horde of joyful SBA questions from Teach Me Anaesthetics. Those links are on the website and in the show notes. Speaking of website, definitely check out GasGasGas.uk for the show notes, diagrams, details, the references. Now we all know guys that this is a bucket of content to consume and it is like drinking from a fire hose. So I want to finish by saying take it day by day. Don’t overcook yourself, don’t freak out and keep sturdying.
Enjoyed this? Review on Apple Podcasts Rate on Spotify
Support the show Help keep the lights on SBA question bank @ Teach Me Anaesthetics

