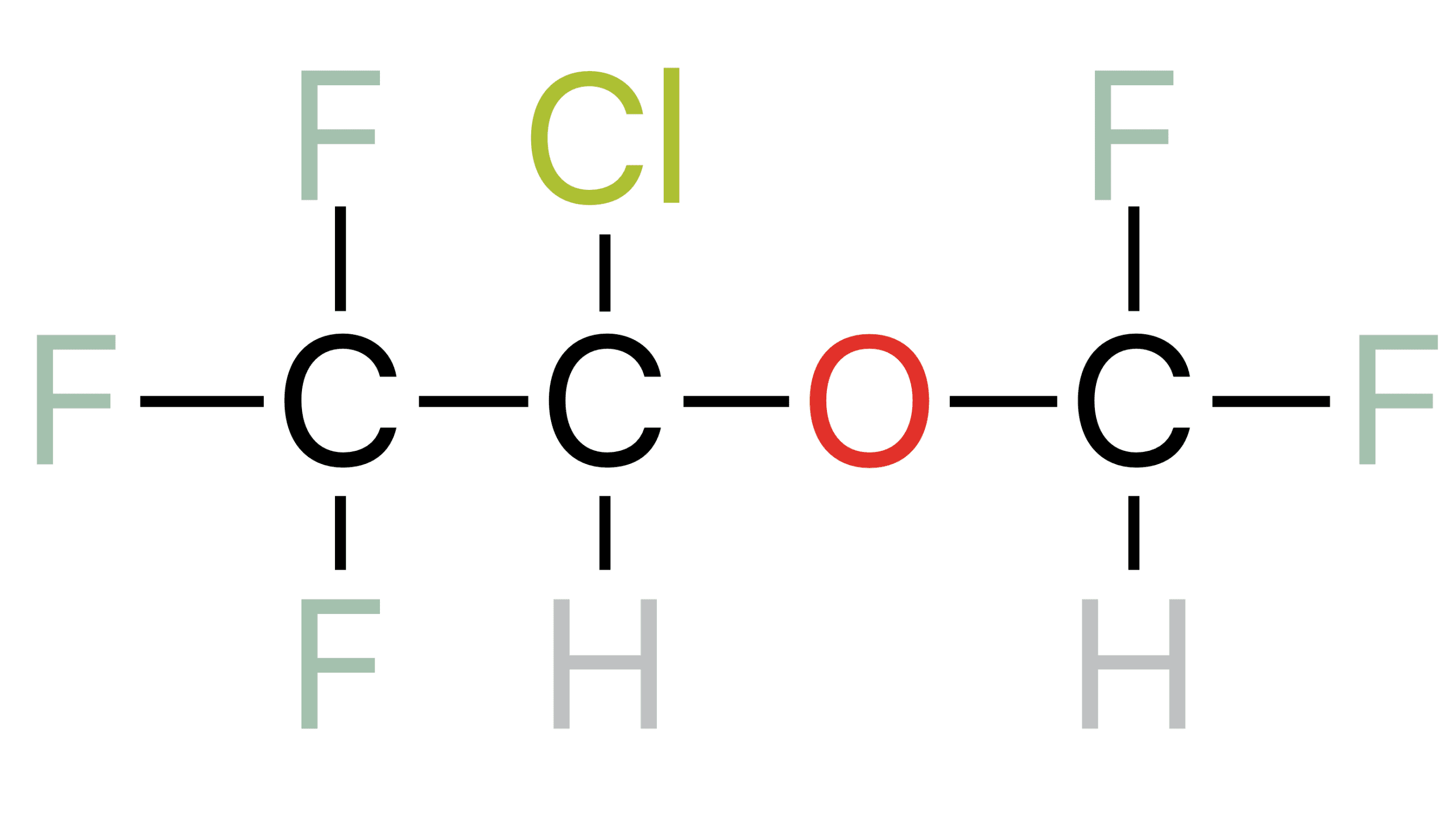Ep.45 – Isoflurane For The FRCA Primary
29 October 2025
Contents
Introduction to the Episode
This episode handles everything you need to know about Isoflurane pharmacology and a few extra bits for curiosity. The physical concept we will focus on in this episode is Daltons Law, which governs the partial pressures of varying molecules/atoms that float around a gas/vapour mix.
This is relevant for anaesthetic agents and also how much oxygen is delivered to your alveoli!
This episode is part of the whole volatile anaesthetic agents series that kicked off the an introduction to volatiles!
Isoflurane Physico-Chemical properties
| Name | Isoflurane |
| Class | Halogenated Methyl Ethyl Ether |
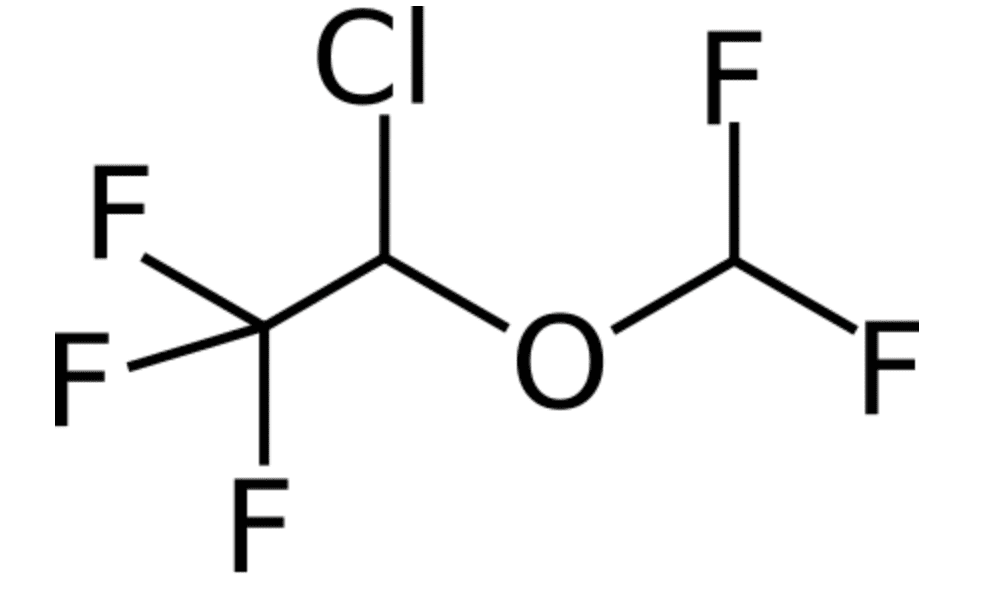
| Chemical Make Up | C3 H2 Cl F 5 O |
| History | Available in UK since 1983, enflurane came about first as easier to manufacture, the three fluorines on the end of the terminal ethyl carbon make it very resistant to metabolism (handy) |
| Isomer Status | Structural Isomer of Enflurane |
| Colour/Appearance | Clear colourless liquid – protected from light. (a Pungent ethereal odour! according to Wade and Stevens) |
| Molecular weight | 184.5 g/mol |
Note the three fluorides at the end, in exams gone by you were asked to draw very common molecules (volatiles, propofol, that sort of thing) maybe they will do that a bit less and focus on clinical relevance a bit more these days… but they still might try a cheeky ‘chemical name’ type question.
The ‘ether’ bit is courtesy of the oxygen molecule binding the two ‘alkyl’ groups.
The carbon with three fluorines bound is the terminal ethyl ether, conferring stability and resilience in the face of ones formidable liver enzymes, and also resulting in no soda lime mischief.
Isoflurane Specific Pharmacological Data
| Isoflurane Boiling Point | 48.5°C |
| Isoflurane Saturated Vapour Pressure (@20°C) | 32 KPa (Peck says 33.2) |
| Mac of Isoflurane | 1.17 |
| Blood:Gas Solubility Coefficient Isoflurane | 1.4 |
| Oil:Gas Solubility Coefficient Isoflurane | 98 |
| ‘Safe’ Isoflurane environmental Exposure | 50ppm |
Isoflurane Pharmacodynamics
| Mechanism of Action | Volatiles seem to disrupt synaptic transmission – especially ventrobasal thalamus Might make GABAa and glycine receptors more jazzed up May antagonise NMDA receptors (N-methyl D-aspartate) ‘Meyer-Overton’ – expansion of hydrophobic regions in the neuronal membrane either within the lipid phase or within the hydrophobic sites of cell membrane proteins ‘Gets around the CNS and causes mischief that renders people hypnotised, amnesic and generally less mobile’ |
| Chief Effect / Actions | Hypnotic |
| Dose | See End Tidal Mac for Dosing |
| Side Effect Profile | |
| Cardio-Vascular Side Effects | Chronotropy : negative effect at high dose Inotropy : negative effect at high dose Dromotropy : Unaltered Lussitropy Coronaries: there has been conflicting evidence for coronary steal, which doesn’t occur in humans, and also ischaemic conditioning that doesn’t bear fruit for homo.anaestheticus At normal doses isoflurane is fairly inoffensive to the CVS Could be described as having a dose related decrease in cardiac output. Vascular Resistance: – Isoflurane is vasodilatory |
| Respiratory Side Effects | Rate – Increased Depth – Decreased Parenchymal effects: > Hypoxic pulmonary vasoconstriction inhibited > Bronchodilating > Airway irritant |
| Central Nervous System Side Effects | ICP : Isoflurane increases ICP Seizure Threshold : Isoflurane is non-epileptogenic Cerebral Vasodilatory Effects (hence ICP rise) Nausea and vomiting |
| Gastro-Intestinal Side Effects | Reduced renal blood flow |
| Metabolic/MSK: Side Effects | MSK: Decreased Tone in a dose dependent manner Can develop a hyperglycaemia + increased GH and Cortisol levels. Insulin is un-altered |
| Toxicity Signs | End point of volatile toxicity: CVS collapse. Malignant Hyperthermia Trigger Note in a toxic collapsed patient, your ability to get the vapours back out of the human is severely impaired…. as they require cardiac output to circulate and excrete agent via the lungs. The difference between anaesthetic dose and CVS collapse dose is wide…. and equipment and monitoring should stop you over doing it. But abroad under a bush with marauding rifle carrying individuals creates a different situation! |
Isoflurane Pharmacokinetics
| Absorption | |
| Distribution | It is expectedly lipid soluble, but less so than [halothane](/episode/halothane/) we know it should have a volume of distribution of sorts… but this isn’t oft quoted! here comes Wissing, Kuhn, Rietbrock and Fuhr |
| Metabolism | Liver Phase I : 0.2% metabolised oxidation / dehalogenation reactions |
Active Metabolites: Other Routes: | |
| Elimination | 99.8% exhaled unchanged |
| Other Notable Information |
Daltons Law of Partial Pressure
John Daltons’ law
Dalton’s law states that the total pressure of a gas mixture equals the sum of the partial pressures of its constituent gases — each component behaves as though it occupies the space alone.
Check out Khan academy for more details on the maths and this Update in Anaesthesia from Dr B Gupta which offers a lovely explainer of most of what you should know for the exams!
All the differing molecules in a gaseous mixture exert a pressure on the ‘fixed volume box’ they are in.
The sum of all these pressures adds up to the total pressure the box experiences.
on the broadest level, your sat on the beach, enjoying the sun (and thus are at sea level)
Pressure at sea level is: 101.325KPa
Conversion are: 1013.25 millibars or 14.7PSI Or 760mmHg Or 1034 CmH2O or 1.01325 Bar or 1 Atmosphere (learn this)
Note: Standard Temperature and Pressure [STP] is 101.325KPa @ 0°C (ie 273.15 Kelvin)
And the components in the atmosphere are
- Nitrogen (78.084%)
- Oxygen (20.946%)
- Argon (0.934%)
- Carbon Dioxide (0.042%)
- And then things like Neon, Helium, Methane, Hydrogen and all sorts of other things that are measured in parts per million
Note that these fractions are quoted for a completely DRY atmosphere….. that is because when you add water vapour the %fractions of other stuff naturally shifts (an maximally humid airdrops pressure marginally as water(mass 18) vapour is lighter than things like nitrogen(mass 28)) .
When there is 4% water in the atmosphere (think a sweltering Thailand you’ve 20.11% O2 floating about) UK normal is 0.25% vapour (more on humidity in a future episode)
To make the numbers less of a headache.
We will assume we are at 100KPa of pressure, And therefore nitrogen makes up 78% of that pressure, an exerts a pressure of 78KPa.
If you were to hoover out everything else in the box except the nitrogen, the pressure in the box would be 78KPa
This 78KPa is the partial pressure of nitrogen in this system.
What does this mean when we add something else to the system? It depends on whether the volume alters to accommodate or not, an unaltered volume with a vapour added to the system will increase the pressure in the system. This is where the ideal gas laws rock up and make themselves known, that is not for today!
If you’ve a fixed 100ml space, and you force in 50ml of isoflurane, you will increase the pressure in the system purely by squeezing a (non-compressible) liquid into the fixed volume, and then when it equilibrates out into its vapour form at what ever fixed temperature you are maintaining this system at, pressure will creep up more as the volatile muscles its way into the space also, and as these agents work to fill as space ‘as if there is no other gas in the space as the molecules do not interact with each other’.
If you are delivering Sevoflurane into your gas mixture in your circle circuit, the pressure increase will be mitigated by stretch in the bag, leak through the APL valve, as well as leak and sampling removal from the circuit (if you turn off a ventilator when on cardiac bypass eventually the bag collapses down as volume lost from circuit exceeds volume added to the circuit)
References
Wade, John G. MD*; Stevens, Wendell C. MD†. Isoflurane: An Anesthetic for the Eighties?. Anesthesia & Analgesia 60(9):p 666-682, September 1981. An excellent and broad article, which covers pretty much all the bases for isoflurane!
https://www.bjanaesthesia.org/article/S0007-0912(17)38798-6/pdf
https://www.bjanaesthesia.org/article/S0007-0912(17)41396-1/pdf
https://www.noaa.gov/jetstream/atmosphere
Thanks for listening. Take it day by day, don't overcook yourself — keep studying.
Transcript
27 min listenRead the full transcript
Gas Gas Gas: Isoflurane – Episode 45
Introduction and Podcast Housekeeping
00:00-01:10
Hello everyone and welcome to yet another episode of Gas, Gas, Gas. You’re perhaps scratching your heads and wondering, have I travelled forward in time? I don’t remember there being an episode last week. And you’re right. After the slight debacle with that opioid quiz episode where the sound went all wrong, and that’s because I was rushing, I’ve decided I’m going to go fortnightly purely so that I can actually focus on delivering something that’s excellent and up to my perpetually escalating standards.
I hope you are sat there and you’re listening carefully. Today we are doing isoflurane, and then we’re going to dabble with Dalton’s Law at the end. We’re going to handle everything you need to know about isoflurane, plus a few extra bits and pieces, just because it’s a drug we probably don’t reach for that often, because we always reach for the yellow sevoflurane-coloured stuff, don’t we? If you missed out the introduction to volatiles episode, I’d say probably wind back and listen to that first so that when I vomit some numbers at you, you’ll know what I’m on about. Get yourself ready for the sevoflurane episode in two weeks’ time, and then it’s going to be halothane.
Classification and Chemical Structure
01:10-02:48
So, classify or die here, folks. What class of drugs is isoflurane associated with? I’m sure you know. I’m sure it’s right on the tip of your tongue. It is a halogenated methyl ethyl ether. I’ll say that again: a halogenated methyl ethyl ether. Rolls off the tongue, doesn’t it? It’s been available for use in the UK since 1983.
Originally, its structurally similar friend, and when I say structurally similar, I mean it’s a structural isomer, so it’s the same molecules in a different arrangement. Its pal enflurane was easier to manufacture, and that’s why enflurane kind of came before isoflurane. They were having some challenges with isoflurane.
What chemicals make up isoflurane? Well, there’s three carbons, two hydrogens, a chloride, five fluorines, and an oxygen molecule. I’d definitely say you need to get on the show notes, folks. I’ve got some images of the chemical structures of isoflurane, and in exams gone by they’ve definitely asked “here, draw isoflurane and sevoflurane and tell us the differences.” Now they might not do that nowadays. They might ask you about propofol because we probably use a lot more propofol, but you probably at least need to have an idea of this, just in case.
One of the important distinguishing factors for isoflurane compared to enflurane is that there are three fluorine molecules on the terminal ethyl carbon. This means that your liver, even though it so desperately desires to break down these molecules because it loves to fight, those fluorine molecules are exceptionally obstructive and very little metabolism of isoflurane occurs.
Physical Properties and Presentation
02:48-04:41
How is it presented? Isoflurane is a clear, colourless liquid. It is protected from light because it doesn’t tolerate light—it breaks down. According to Wade and Stevens, referenced in the show notes, it has a pungent ethereal odour.
Now, interestingly, we all reach for sevoflurane for induction of anaesthesia in the gas induction variety. But you can also do it with isoflurane. The trick with isoflurane, seemingly, is that you can’t just smash it to eight and hold that mask on that person because then it is irritant. But if you sneak it in slowly but surely in the gently, gently, softly, softly style, then you can use isoflurane as a gas induction agent, which worked better than enflurane and worked better than diethyl ether that was also available floating around in the cupboards once upon a time.
Its molecular weight is 184.5 grams per mole, and its colour coding is purple. These are important if you get asked in an exam about how to avoid putting the wrong vapour agent in the wrong vaporiser—there’s colour coding. The bottle to vaporiser interface is keyed, so you cannot inadvertently attach one to the other. Once upon a time you used to be able to just pour it in. You can’t anymore. And this is important because each vaporiser has a slightly different set of thermal compensation and expectation of the saturated vapour pressure inside the vaporiser with which to then deliver a percent fraction into your gas flow. So if you mix it up, you can cause mischief.
Key Pharmacological Numbers
04:41-06:14
With all the volatiles, at least the ones that we use commonly, and I would argue that that is going to be isoflurane, sevoflurane, and they’ll probably have the potential to ask you about desflurane, you should know sets of numbers. Obviously this really helps you treat patients with compassion and care, but eventually it will mean that you have in your head a frame of reference as to which one behaves more like this and which one behaves less like this.
So isoflurane boils at 48.5 degrees. Now, unless we are unfortunate and doing anaesthesia in the Sahara Desert, in our cold and gloomy United Kingdom climate, that means the saturated vapour pressure for isoflurane remains predictable at standard hospital temperatures. The saturated vapour pressure at 20 degrees of isoflurane is 32 kilopascals, unless, of course, you’re reading Peck and Hill, who seem to have a different opinion and say it is 33.2 kilopascals. Great.
Remember folks, saturated vapour pressure is the partial pressure that isoflurane will exert once it has reached equilibrium between the liquid and the vapour states at 20 degrees.
The MAC of isoflurane is 1.17. So generally aim for an end-tidal of 1.2 and you’ll have a MAC of one when you’re using isoflurane.
Blood:gas solubility is 1.4. Remember sevoflurane is 0.69, and desflurane, if I’m remembering correctly, one second, is 0.45.
Blood:Gas Solubility and Clinical Implications
06:11-08:18
So you need to use your frame of reference for this. The higher the blood:gas solubility is, the better at dissolving that gas is in that blood. So actually, isoflurane is fairly dissolvable. What does that mean? Well, eventually, the amount of isoflurane that ends up in your brain dictates when you go to sleep and when you wake up. But isoflurane has to make it from your anaesthetic circuit to your alveoli, then across the membrane and into the blood that’s circulating, before it then gets to the brain where it can be effective.
So the better it dissolves into blood, the better your blood is able to act like a buffer, which means that with a high blood:gas solubility, technically there’s a slower onset when you compare it to sevoflurane or desflurane. Why does that matter clinically? Well, number one, does it matter clinically? Some might say arguably not. It does mean that if I need to raise or lower my volatile in response to the patient’s consciousness, it’s a little bit more sluggish, compared to sevoflurane and definitely compared to desflurane. If I were to go and interview your patient the next day and they told me their experience of having your anaesthetic was delightful, then probably doesn’t matter. But if you need to be nimble, then you will feel less nimble when you use isoflurane.
The oil:gas solubility is 91. I’m hoping I’ve not confused you. So there is a blood:gas solubility coefficient, which is 1.4. There’s also an oil:gas solubility coefficient, and this is used as a surrogate for the lipid solubility of the drug because, at the end of the day, your membranes are fatty and lipophilic. So the better it dissolves in oil, the more potent the drug can be. So isoflurane dissolves fantastically in fat and oil, and sevoflurane is 47, so sevoflurane is about half.
And I think desflurane is about 27, 28, somewhere in that region. So of those three drugs, isoflurane is the most fat-soluble. What does that mean clinically? Well, it means that isoflurane is a very potent drug because it’s so fat-soluble. So you don’t need very much end-tidal concentration of isoflurane to put someone to sleep—1.17% or 1.2%. So it’s very, very useful from that perspective.
Partition Coefficients and Distribution
08:18-10:45
The brain:blood partition coefficient is a rarer, a more obscure parameter than blood:gas solubility or oil:gas solubility. And brain:blood partition coefficient for isoflurane is 1.6. So sevoflurane is 1.7, desflurane is 1.3. Now brain:blood partition coefficient is kind of pointless actually for inhalational anaesthetics because it’s always about 1 in that range anyway. Saying the brain:blood partition coefficient is one higher is not saying that it’s better at distributing to the brain—it’s saying it’s a tiny bit better, maybe. What matters more is that blood:gas solubility.
Isoflurane has a blood:muscle partition coefficient of 2.9. So it distributes preferentially and nicely into your patient’s muscle. Now muscle is about 35% of your body weight as a 70kg patient. So isoflurane is going to be quite nicely distributed when the anaesthetic has been ongoing for a while. And then it’s going to spend a little while slopping out of that muscle when the anaesthetic has stopped, or when you’ve spun off your volatile. Sevoflurane is 2.7, desflurane 2.0 for muscle.
And then the third thing to tell you that you should note is that the blood:fat partition coefficient for isoflurane is 45. Sevoflurane is 47.5, desflurane is 27. So a patient who is more obese where we’re using isoflurane, then over time isoflurane would theoretically distribute nicely into fat and build up in that fat. Fat is roughly 25% of the 70kg patient—that’s the presumption at least. But in the obese patient, obviously it’s much larger. So therefore, theoretically, that drug is going to hang around a bit longer on recovery in someone who is obese. But in reality, there’s no difference between obese and non-obese patients in the recovery from isoflurane.
MAC Values and Factors Affecting MAC
10:45-13:54
So we’ve talked about potency. Potency is inversely related to MAC. I’m going to have to get that T-shirt made for that one. So the smaller the MAC value, the less concentration you need to anaesthetise a patient, the more potent the drug. So the more potent the drug, the smaller the MAC. Inversely related. If I had to teach you, write these drugs from most potent to least potent, it’s going to be isoflurane, then sevoflurane, then desflurane. And then actually, the order doesn’t change when you compare it to enflurane and halothane. Halothane is about 0.75, and enflurane is about 1.7. So halothane is the most potent, and then going up the stack it’s enflurane, which is the least potent. That’s just a nice frame of reference, right?
The important thing, of course, is MAC. MAC is the minimum alveolar concentration required to prevent movement on a skin incision in 50% of patients. And it’s used as a measure to compare these drugs. So you’re effectively saying, “This is the concentration in alveoli that would correlate with a plasma concentration that is related to the concentration in the brain that’s related to an amount of drug that means 50% of my patients don’t move at incision.” So you give it to 100 patients, 50 move, 50 don’t. MAC of one is when 50% move. So ideally you want to be aiming for higher than a MAC of one because you want everyone to not move.
Probably about a MAC of 1.3 is what we target when we’re delivering anaesthesia. And then the concept of MAC-awake exists, and this is the concentration at which 50% of your patients open their eyes to command. So that’s approximately 50% of MAC. The MAC that gives you a 95% chance that your patient doesn’t move to incision is called ED95, the effective dose in 95%—that’s 1.2 to 1.3 MAC. And then MAC-BAR, which is the lowest level that prevents autonomic responses to noxious stimulation, is 1.5 to 2 MAC.
Now these are important concepts because you will get asked to define them in the exam, but also it puts into context what you’re actually doing. So for an anaesthetic where someone’s going to get cut in half for bowel surgery, you’re probably going to need a decent amount of volatile. And it demonstrates in part why we use analgesics—because that reduces the amount of volatile that’s required.
Factors that affect MAC—well, there’s things that are consistent and there’s things that vary. So importantly, if you’ve got a patient who’s exercised loads today, they’ve run a marathon and they’re depleted and they’re exhausted, that doesn’t affect MAC. There’s no link between MAC and physical fitness. There’s no link between MAC and gender. There’s no link between MAC and thyroid function. There’s no link between MAC and hypo- or hypercarbia. There’s no link between MAC and chronicity of anaemia, which is quite interesting. There is obviously a link between potassium and anaesthesia because potassium affects depolarization and repolarization, but it doesn’t affect MAC.
Okay, so what about the things that do reduce MAC? Acute intoxication reduces MAC. So if you’ve got a patient who rocks up absolutely hammered, they’re going to need less volatile to keep them asleep. Chronic alcohol excess increases MAC, so you need more volatile. Pregnancy reduces MAC by about 30% to 40%, so you need less volatile in an anaesthetic. Acute blood loss can reduce MAC. Alpha-2 agonists reduce MAC. Opioids reduce MAC. Benzodiazepines reduce MAC. Lidocaine reduces MAC. Propofol reduces MAC. You get the picture—other drugs that we use for anaesthesia reduce MAC.
Metabolic changes—so hypothyroidism, hyponatraemia, all these sorts of things reduce MAC. Extreme age, so neonates and the very elderly, will reduce MAC. And then your extremes of temperature—hypothermia reduces MAC and hyperthermia reduces MAC. So a patient who’s really, really hot going for surgery, very high temperature, will need less volatile.
And then there are things that increase MAC. Young age increases MAC—so children will need more volatile. Chronic alcohol excess. Amphetamines and cocaine—so your recreational drug use—increase MAC. Hypernatraemia increases MAC.
Pharmacodynamics: Cardiovascular Effects
13:54-15:45
So what does isoflurane actually do? What’s its pharmacodynamics? What does this drug do to the body? Starting with the cardiovascular system. Well, isoflurane is a direct vasodilator. It lowers the systemic vascular resistance. Isoflurane has no effect on heart rate. It has no effect on cardiac output, so your stroke volume and your heart rate stay consistent. But because it lowers the systemic vascular resistance through that peripheral arterial vasodilation, it reduces your blood pressure. And that’s the main cardiovascular system effect that you’ll notice with isoflurane when you’re using it on a patient.
The consequence of this is that because isoflurane is dilating all the vascular beds, that includes your coronary vascular beds. It dilates the coronary arteries. And a consequence of that is that in coronary steal syndrome, which is this delightful syndrome where because you’ve got poorly perfused myocardium, if you dilate everything else, then the well-perfused areas perfuse even better and the less-well-perfused areas get even less blood flow. And so back in the day, we used to worry about this with isoflurane. But in reality, it’s not a thing. Isoflurane is fine to use in patients who have coronary artery disease. Historically we’ve been worried about it. Nowadays we don’t worry about it.
You can see coronary steal if you measure coronary sinus blood flow, but in reality, there’s no evidence that it causes myocardial ischaemia in any of the papers that are available or any of the trials that are available on the topic. So you should definitely mention it if you’re in the exam because it’s a thing and it’s been examined before, but simultaneously recognize that it’s not a problem when you just blithely use isoflurane on people. So isoflurane causes vasodilation. It might vasodilate the coronary arteries a bit more than others, but that doesn’t make it a problem clinically.
Isoflurane is also negatively inotropic, so it reduces myocardial contractility. It causes dose-dependent myocardial depression, but it’s a much weaker negative inotrope than either halothane or enflurane. So if I think about volatile agents, the ones that are not so good for inotropy are halothane and enflurane, and isoflurane is actually pretty reasonable in that regard.
Pharmacodynamics: Respiratory Effects
15:45-16:35
Respiratory system: isoflurane reduces respiratory rate, it reduces tidal volume, and therefore it reduces minute ventilation. As a consequence of that, your PaCO2 rises. And then because your central chemoreceptors that sense a rising PaCO2 are partially attenuated by the isoflurane being on board, you tolerate that increase in PaCO2. So that’s your respiratory changes with isoflurane on board. Because there’s this reduction in tidal volume, it predisposes patients to atelectasis if you’re using a volatile-based anaesthetic for a lengthy surgical procedure.
Isoflurane, similar to sevoflurane, is a bronchodilator. I suppose it makes sense—you’re dilating all of the vascular beds, you’re also going to dilate the bronchioles. So isoflurane is actually a reasonable choice in the patient who has reactive airway disease. Similarly, because it is a bronchodilator, if you’re going to be reaching for your bronchodilator treatment pathway, then dialling up the volatile might be one of those tools in your toolbox.
Pharmacodynamics: Central Nervous System Effects
16:35-17:17
CNS-wise, isoflurane reduces cerebral metabolic rate. It actually does this more than sevoflurane, and it does it more than desflurane. So isoflurane reduces your CMR. It reduces your cerebral metabolic rate of oxygen. The consequence of that reduced CMR is that the brain doesn’t need as much substrate because it’s doing less. Your cerebral blood flow then falls in response to that. So with isoflurane, your cerebral blood flow will come down as a consequence of the lower CMR. But up to one MAC, your cerebral autoregulation is preserved, so the brain is still able to autoregulate blood flow to itself based on its needs up to a MAC of one. When you get beyond that, then the brain loses its ability to autoregulate, and cerebral blood flow is then dictated by the mean arterial blood pressure.
Isoflurane impairs hypoxic pulmonary vasoconstriction at about 1.1 MAC—so similar to sevoflurane, similar to desflurane. Interestingly, propofol also impairs hypoxic pulmonary vasoconstriction. This means that in a patient who’s got a poorly ventilated area of lung, your body doesn’t shunt blood away from that area as effectively. And so theoretically you’d have slightly worse gas exchange, slightly lower PaO2. But in reality, it doesn’t make a massive difference clinically because there’s so many other compensatory mechanisms going on.
Pharmacodynamics: Other System Effects
17:17-17:51
Isoflurane provides some degree of muscle relaxation and it potentiates neuromuscular blockade. So you’re going to need less of your muscle relaxant when you’re using isoflurane. All the volatiles do that to some degree.
Isoflurane is a trigger for malignant hyperthermia. It is not appropriate to use in patients with a susceptibility to malignant hyperthermia.
With regards to your hepatic and renal function: isoflurane causes some reduction in renal blood flow, but importantly there’s minimal metabolism of isoflurane, so it doesn’t produce problematic metabolites. We’ll talk about that in the pharmacokinetics section.
Isoflurane doesn’t affect intraocular pressure significantly. And in terms of uterine tone, it causes some uterine relaxation at higher concentrations—so it’s tocolytic at higher doses. For obstetric anaesthesia at low doses, it’s fine, but you wouldn’t want to crank it up really high.
Isoflurane does not cause seizures. In fact, isoflurane has anticonvulsant properties. So if you’ve got a patient who has epilepsy or who’s at risk of seizures, isoflurane is not going to be a problem. Contrast that with enflurane, which can cause seizure activity at high doses. That’s not an issue with isoflurane.
Clinical Use and Monitoring
17:17-17:48
One of the challenges with isoflurane, and the challenge with all the volatiles, is that if for whatever reason your end-tidal gas monitoring wasn’t working, and let’s say you’ve got a patient who’s having a ketamine TIVA and you’re supplementing it with isoflurane—probably don’t use isoflurane, ketamine is a good choice. Then you might find yourself in a situation where you don’t know how much you’ve dosed the patient with and where you are. And that’s why modern anaesthesia is super duper safe, and historic anaesthesia where we didn’t have clever gas analysers and all these sorts of things was less safe.
Pharmacokinetics
17:48-18:44
Okay, so what about the pharmacokinetics of isoflurane? And this is pretty sparse, I’m afraid, guys. If you get asked what the pharmacokinetics of isoflurane are in the exam, remember this is what the body does to the drug.
As you’d expect, isoflurane is quite lipid-soluble, so it distributes eventually quite evenly. It will preferentially distribute to areas of high cardiac output before equilibrating throughout the patient as you journey through your anaesthetic.
Absorption: well, it’s readily absorbed through the alveoli. It’s kind of a given, but maybe you should open with “it is readily absorbed through the alveoli.”
Metabolism: this is something that’s important with volatiles. Isoflurane is 0.2% metabolised via oxidation and dehalogenation reactions. Therefore, you can say that it is 99.8% eliminated unchanged via the lungs.
And boom, that is everything you really need to know about isoflurane and a few little extra bits here and there.
Support the Show
18:44-19:09
So well done guys for getting this far. I’ll just take the cheeky opportunity to say if you’re enjoying the show then feel free to fire off a donation. The link’s in the show notes, helps keep the show running. Or check out the link to Buy Me a Coffee that Dr. J Toe suggested I should do because it’s perhaps a bit simpler. Maybe I was being awkward by using PayPal. So yeah, there’s a Buy Me a Coffee link now as well, folks.
Dalton’s Law of Partial Pressures
19:09-23:04
So you’ve had some drug teaching. What about some useful physics-y type teaching? We need to think about Dalton’s law of partial pressures. This is one of a multitude of laws that you need to get a grip of to understand how gases behave in order to bag points in the exam, ultimately.
Dalton, or John Dalton, Dalton’s law states that the total pressure in a system is going to be the sum of the partial pressures of each individual gas inside that system. So if we imagine a box, and in that box the pressure is 100 kilopascals, and in that box we have 21% oxygen and 79% nitrogen, we know that therefore there’s a 21 to 79 split. That means oxygen is generating the equivalent of 21 kilopascals of pressure, and the nitrogen is delivering 79 kilopascals of pressure.
Now if you were to remove the oxygen from that system, the pressure in that system becomes 79 kilopascals. This works for ideal gases because the presumption is that these gas particles are so far apart in this lovely gaseous space that they are not interacting with each other.
Unfortunately, in our awkward world of presumably and apparently metric atmospheric pressure, one atmosphere is the equivalent of 101.325 kilopascals when we’re thinking about sea level, which is what one atmosphere is. There are conversions between all these various pressures that you’re painfully going to have to know for the exam, so that you can, in the exam, decide to convert millimetres of mercury to centimetres of water or an atmosphere to a bar to kilopascals to pascals, et cetera, et cetera.
Why do we care about Dalton’s law of partial pressures? Well, we mentioned the saturated vapour pressure of isoflurane earlier. And this bears out: if we’ve got that delightful system of oxygen and nitrogen, and then we put some isoflurane vapour in there as well. Squirt that vapour in, or in reality, we tip some vapour in the bottom of the bucket and we will decide to presume that the volume of liquid we’ve added is irrelevant. It equalises, it vaporises, it reaches its saturated vapour pressure.
Then you have squeaked in, because none of that oxygen or nitrogen has escaped, another 32 kilopascals of pressure into that system. So now the box, because that stuff’s all vaporised, is operating at a higher pressure because it’s muscled its way in.
Yet another important pressure-y, physics-y type thing to remember is this concept of standard temperature and pressure, which is sometimes quoted or sometimes examined. And this is atmospheric pressure, i.e., 101.325 kilopascals, but at zero degrees C, which is the equivalent of 273.15 Kelvin. This is sometimes also used when you’re being delightfully examined on these principles.
Eventually we’re going to have to talk ideal gas laws. There is a Vivacast on that that covered it, but we’ll recover these things because you need to—I think I’ve read about the ideal gas laws like fifty times, because it always falls out of your brain. And now that Charles is king, well, we’re going to have to change the whole mnemonic for remembering it. Very unfortunate. Sure, there’s a slightly questionable one you could imagine with one of our non-members of the royal family, but unfortunately is a member of the royal family, who’s bringing the whole thing into disrepute. I’ll have a think about that.
So, wanted to be reasonably brief on Dalton’s Law. There are a number of links and references in the show notes that would explore that and explain that in further detail if you’re interested, as well as another example of how the system might behave when you tip isoflurane into a bucket that is in fixed volume and actually considering the pressure alteration that that isoflurane might cause.
Summary and Conclusion
23:04-24:20
So, what we covered in this episode: we’ve compared isoflurane with sevoflurane and desflurane, and we now know that isoflurane is quite a potent drug. You don’t need very much of it to achieve anaesthesia. It gets out to the blood slower, technically, than sevoflurane and desflurane. But again, it’s a nice stable drug that is very minimally metabolised.
We’ve touched on Dalton’s law of partial pressures, courtesy of John Dalton. John Dalton, having been born in the year 1766, and considered a chemist, physicist, and meteorologist—a fellow with many feathers in his cap. And I’ve told you that you unfortunately need to learn how to convert different pressures, be that millimetres of mercury, centimetres of water, bar, kilopascals, and atmospheres. If you can do that, there will very likely be a question on that in the exam because it’s just something that’s quite examinable. So yeah, learn that. There’s a list of it on the show notes as well. Fun times!
Anyway, I hope you have a nice weekend. I hope you’re super excited to learn more about sevoflurane. I’ve learned a few things about sevoflurane whilst preparing for that episode. For those who are also studying for the primary FRCA viva, keep at it, keep going. The end will soon be nigh. Try and practise with your friends as much as you can, even if you feel awkward.
Enjoyed this? Review on Apple Podcasts Rate on Spotify
Support the show Help keep the lights on SBA question bank @ Teach Me Anaesthetics