Introduction
Cisatracurium is one of the ten isomers found in Atracurium, and it has been selected out as a useful, potent, predictably degrading drug with a very stable side effect profile!
Althought, it might be that, whilst Cisatracurium probably sounded like a great idea at the time… it was perhaps batting for a marketing USP that wasn’t quite there..
If we were being particularly clever we could call it the R-cis-R’-cis isomer of Atracurium besilate, it is 3 times as potent as the Atracurium mix in your anaesthetic room fridge
Recovery time is faster than vecuronium, and even quite large doses maintain a stable cardiovascular system. One of its key aims was a non-histamine releasing benzylisoquinolinium thats offset was independent of the hepatic and renal systems.
Article Spring Board
Cisatracurium Pharmacology
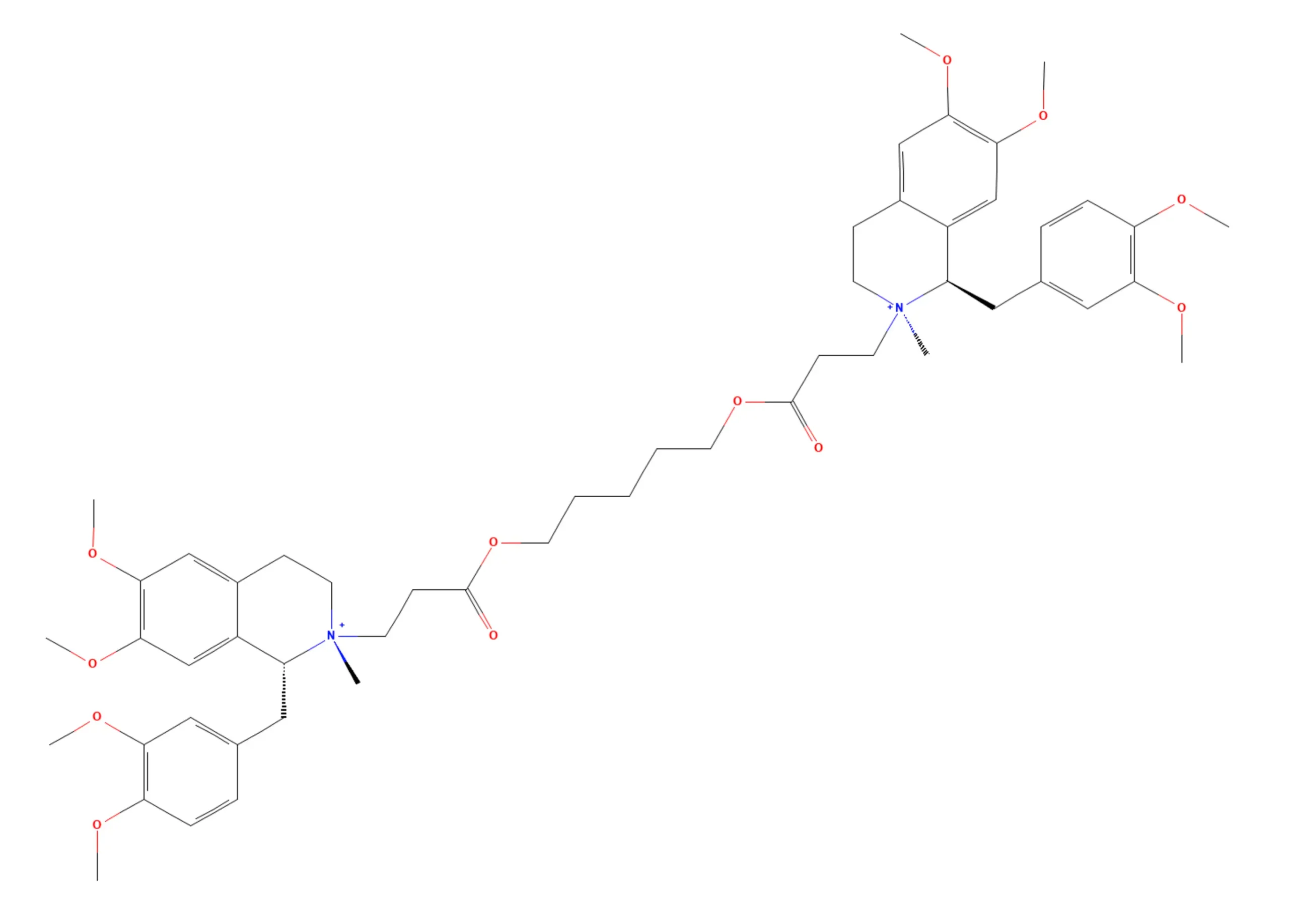
Cisatracurium Physico-Chemical properties
| Name | Cisatracurium besilate – brand name ‘Cisblok!’ and Nimbex (excellent neuromuscular blocker) |
| Class | Bis-Benzylisoquinolinium Ester (one of 10 diastereoisomers of atracurium) Non-depolarising, competitive NMB. (Dia-Stereo-Isomers refer to isomers that having multiple chiral centres can have parts that appear a mirror image of another molecule and other parts which do not) |
| Chemical Make Up | C53H72N2O12+2 |
| History | According to exceedingly reliable resources on the internet… Burroughs Wellcome co. of Dartford in the UK, held the license for atracurium from Strathclyde University set about testing its individual isomers, cisatracurium was first synthesised in 1989, burroughs wellcome is now part of glaxosmithkline. |
| Isomer Status | One of Ten! |
| Colour/Appearance | Clear Colourless or pale yellow aqueous solution in 5,10,20ml vials Containing 6.7mg of Cisatracurium besilate – equivalent to 5mg/ml Cisatracurium Needs refrigeration at 2–8 °C pH 3.25-3.65 (quite acidic!) |
| Incompatibilities | Acidity means it does not play well with barbiturates, propofol and ketorolac |
| Molecular weight | Cisatracurium = 929 g/mol (if you decide to count its besilyate salt, then 1243.5 g/mol) (Miv 1029.3, roc 529, sux 361, trac 929) |
This is Cisatracurium, i’m sure all FRCA examiners can spot the differences!
Cisatracurium Pharmacodynamics & Side Effects
| Mechanism of Action | Competitive non-depolarising neuromuscular blockade (Check out rocuronium for more details) |
| Chief Effect / Actions | Antagonism of acetylcholine at the nicotinic (N2) receptor situated on the post synaptic membrane of the skeletal neuromuscular junction (nAChRs) |
| Dose | Intubating: 0.15mg/kg (often the quoted intubating dose of a NMB is 2-3x ED95) Maintenance: 0.03 mg/kg Infusion: 1.2-1.5 micrograms/kg/min in theatre : more req in ICU 2.6-3.2 mics/kg/min) Onset: More = faster onset – 0.1mg/kg 4.6-5.8mins, whereas 0.15mg/kg=2.4-2.7mins ‘good to excellent intubating conditions after 120sec @ 0.15mg/kg’ 0.2mg/kg achieves decent conditions in 90s Offset: (more drug = lasts longer) 0.1mg/kg = 33-45mins, 0.15mg/kg= 55mins (for 25% of twitch to recover) ED95 for paralysing dose : ~0.05mg/kg (to produce 95% twitch suppression) like other agents, once recovery begins its progression takes a similar amount of time regardless of dose. Neostigmine accelerates this return of power |
| Cardio-Vascular Side Effects | Headline : MAP / HR stable with rapid and large boluses. (<8x ED95) 0.4% bradycardia |
| Respiratory Side Effects | Rate: Paralytico Depth: Paralytico! Parenchymal effects: 0.2% bronchospasm |
| Metabolic/MSK: Side Effects | 0.1% rash 0.2% flushing |
Cisatracurium Pharmacokinetics
| Absorption | n/a |
| Distribution | Volume of distribution 0.11-0.16 L/kg at steady state as infusion. Crosses the placenta (try and give the Atracurium if you need to post delivery to facilitate closure) Less protein bound than Atracurium – Deranged Physiologysuggests 38% – studies note that because of its spontaneous degradation achieving these measurements is not without challenge. Peck and Hill suggest 15%! (table 11.5, section II, core drugs in anaesthetic practice) |
| Metabolism Remember: Phase I : [oxidation, reduction, hydrolysis] (more cytochrome action here) (more O2 needed) occurs in inner aspect of liver acinus… Phase II: [conjugation, glucoronidation, acetylation, sulphylation] (less O2 needed) occurs in outer aspect of liver acinus… | Prime : Hoffman Degradation (Cleaved link between quaternary nitrogen and central chain) Second : Hydrolysis by non-specific esterase (but significantly less as a fraction compared to Atracurium) Inactive Metabolites Hoffman : Laudanosine + quaternary monacrylate. Laudanosine is metabolised by the liver and its metabolite is really excreted. Hydrolysis: yield a Quaternary alcohol and an acid. |
| Elimination | Half-life : 22-35 mins Clearance : 4.6 to 5.7 ml/min/kg 4% feces 10-15% unchanged in urine |
Cisatracurium in Anaesthesia
Why pick Cisatracurium?
Its onset time and duration of action is intermediate in the spectra of muscle relaxants, well it causes less histamine release, but if you were to pick up a amino steroid, you would also avoid this.
It breaks down via Hoffman degradation, plus a smaller esterase component, so in a histamine risky patient who also has knackered liver/kidneys you might be tempted.
What is a ‘histamine risky patient’? Perhaps someone with severe asthma, or who has a high propensity to hives when exposed to random allergens at home.
There is a line of reasoning that it might be tempting in critical care environments, as the breakdown product of this benzylisoquinolinium is laudanosine, which in high plasma concentrations, in animal models has been found to produce seizures (@>than 17,000 micrograms/L). Given Cisatracurium is more potent, less is required as an infusion, leading to lower levels of laudanosine.
The trouble is, there is yet to be demonstrable evidence that humans have achieved such levels of Laudanosine – but it might be a consideration in a brain injured/status epilepticus + severe acute respiratory distress type patient – but this is quite a niche case!
Comparative Potencies
Taking atracurium as your baseline, Cisatracurium is 3x the potency, vecuronium 4-5x the potency whereas rocuronium is 1.3x less potent.
We know rocuronium is less potent, requiring larger doses, but this is clinically useful as you will achieve a greater concentration gradient plasma:effect site as well as delivering more agent to said effect site, the so called Bowmans principle (potency is inversely related to onset), leading to faster onset times relatively.
Laudanosine
Breakdown product of benzylisoquinolinium metabolism, cleared by the liver.
- A single adult dose of cisatracurium might yield 20-38 microgram/litre plasma conc
- A single adult dose of atracurium yield 190-223 micrograms/litre laudanosine.
- Continuous infusion CisAtracurium:Atracurium 210-1260 micrograms/litre : 520:8650 micrograms/litre respectively.
Levels in a human, in studies found to hint 520-8650mcg/litre plasma. (table 3 Bryson paper)
Potentiating NMBs
Remember that there are many ways to impair the function of skeletal muscle!
Depending on site of action:
- Blockade of ACh synthesis = Hemicholinium
- Depletion of ACh stores = Tetanus Toxin
- Prevention of ACh release = Gentamicin, magnesium
- Nicotinic Receptor Blockade
4.1 – Depolarizing blockade of AChR
4.2 – Non-depolarizing blockade of AChR
Volatiles – there is an isoflurane binding site on the nicotinic AChr, clever folks have demonstrated that an NAChR with isoflurane bound results in inhibited ion inflow through the channel. Approximately Mac 1.7 iso 1.3 sevo are equivalent. you’ll see a change in twitches from your cruising altitude 1 Mac vs your 0.5 waking the up best check the twitches again moment pre neostigmine.
I wonder if TOF reversal parameters should differ between a volatile and TIVA anaesthetic??
Lithium, which reduces MAC requirements is also synergistic with neuromuscular blockade, and this is because of its ‘softening up’ of the polarity of excitable tissues. Lithium can edge into cells via sodium channels, however cells are note generally very good at pumping it back out again. Potassium is displaced in order to counter this excess of positively charged intracellular ion and this leads to a membrane that is less negative (when we consider electrical potential as the amount of negative outside vs inside of a cell)
Patient insensitivity to NMBs
Carbamazepine and phenytoin + other AEDs can make a patient resistant to NMBs, there are a number of factors that lead to this effect:
- Enzyme induction leading to faster metabolism of drug (Aminosteroid NMBs like Vecuronium/rocuronium)
- Increased production of alpha1 acid glycoprotein (phenytoin/phenobarb) reducing the unbound fraction of atracurium
- Direct Neuromuscular changes, AEDs are membrane stabilising (as you would expect as they reduce seizures)
- A metabolite of carbamazepine is a NMB blocker!
Don’t forget that acute admin of AEDs (like your new seizure patient) will need less anaesthetic because of the keppra etc. but that your chronic patient needs more!
Dantrolene (intracellular skeletal muscle relaxant antagonising ryanodine receptors) potentiates NMBs.
nb ryanodine receptors are high conductance L-Type calcium channels on the sarcoplasmic reticulum that release calcium into the sarcoplasm. (but they are also found in multiple other cells too)
Baclofen – GABAB Agonist, modulating skeletal muscle tone favourably reducing spasticity – not directly synergistic with NMBs.
Don’t forget patients with a surfeit of additional nicotinic acetylcholine receptors courtesy of significant burns or denervation they may require greater doses of non-depolarising neuromuscular blocking agents.
And Myasthenia Gravis patients mixed with suxamethonium fair poorly, as you are potentially unable to fully depolarise their muscle with there paucity of nAChRs!
Patient NMB sensitivity
If we think in a bit more broadly, there are other patients who are ‘more susceptible’ to neuromuscular blocking drugs, and we should be considered in our application, this is a fair list, and something that is relevant in life, and in the final FRCA exams.
- Neuromuscular dysfunction patients:
- ALS / Motor Neurone disease
- Myasthenia Gravis
- Guillain-Barre Syndrome
- Myotonic Dystrophy
- Muscular Dystrophy (duchenne)
- Auto-Immune:
- Systemic lupus erythematosus
- Polymyositis
- Dermatomyositis
- Other:
- Don’t forget to think about burns patients, cerebral palsy patients and hemiplegic patients.
Ultimately it’s important to check the ToF! weight up the odds of interaction, and if in doubt, Rocuronium + sugammadex or avoidance of neuromuscular block facilitated by sensible quantities of remifentanil if needed is a worthwhile escape!
Summary
References
- Bryson HM, Faulds D. Cisatracurium besilate. A review of its pharmacology and clinical potential in anaesthetic practice. Drugs. 1997 May;53(5):848-66. doi: 10.2165/00003495-199753050-00012. PMID: 9129870.
- Szakmany T, Woodhouse T. Use of cisatracurium in critical care: a review of the literature. Minerva Anestesiol. 2015 Apr;81(4):450-60. Epub 2014 Apr 10. PMID: 24721895.
- Kumar, Vasam Rajesh, Sanjot Ninave, and Vivek Chakole. 2021. “Pharmacology of Cisatracurium Besylate”. Journal of Pharmaceutical Research International 33 (60B):1701-9. https://doi.org/10.9734/jpri/2021/v33i60B34796.
- Paul, Matthias MD, DEAA,; Fokt, Ralf M.,; Kindler, Christoph H. MD, DEAA†,; Dipp, Natalie C. J.‡,; Yost, C. Spencer MD*. Characterization of the Interactions Between Volatile Anesthetics and Neuromuscular Blockers at the Muscle Nicotinic Acetylcholine Receptor. Anesthesia & Analgesia 95(2):p 362-367, August 2002. | DOI: 10.1213/00000539-200208000-00022
- Harbell MW, Dumitrascu C, Bettini L, Yu S, Thiele CM, Koyyalamudi V. Anesthetic Considerations for Patients on Psychotropic Drug Therapies. Neurol Int. 2021 Nov 29;13(4):640-658. doi: 10.3390/neurolint13040062. PMID: 34940748; PMCID: PMC8708655.
- Lithium: mimicry, mania, and muscle relaxantsFlood, Simon et al.Continuing Education in Anaesthesia, Critical Care and Pain, Volume 10, Issue 3, 77 – 80
- Soriano, S.G., Martyn, J.A.J. Antiepileptic-Induced Resistance to Neuromuscular Blockers. Clin Pharmacokinet 43, 71–81 (2004). https://doi.org/10.2165/00003088-200443020-00001
- Martyn JA, White DA, Gronert GA, Jaffe RS, Ward JM. Up-and-down regulation of skeletal muscle acetylcholine receptors. Effects on neuromuscular blockers. Anesthesiology. 1992 May;76(5):822-43. doi: 10.1097/00000542-199205000-00022. PMID: 1575351.
“Thanks for listening guys… Every day you are getting better at this. Take it day by day, don’t overcook yourself, don’t freak out, and keep studying!”
Podcast Information
Listen to the Show On
Contact & Feedback
- Comments: Share your clinical experiences and ask questions!
- Corrections: Help us improve accuracy and clarity
- All of Toms’ Fan mail will get forwarded
- Email Us Here
Follow GasGasGas On
- FaceBook: Facebook – Gas Gas Gas
- BlueSky:Gas Gas Gas (@gasgasgaspodcast.bsky.social)
- InstaGram: GasGasGas
- X / Twitter: GasGasGasFRCA (@GasGasGasFRCA)
Transcript – Cisatracurium: Pharmacology, Pharmacokinetics & Neuromuscular Junction
Gas, Gas, Gas – FRCA Primary Exam Podcast | Episode aired: Tuesday 17th February
Introduction and Episode Overview
Key points:
- Cisatracurium pharmacology, pharmacokinetics, and pharmacodynamics
- Comparison with atracurium and other neuromuscular blocking drugs (NMBDs)
- Potentiation and inhibition of NMBDs
- Structure and function of the neuromuscular junction (NMJ)
[00:00–01:25] Hello, Team Anaesthesia. Welcome to Gas, Gas, Gas – the best anaesthetic science podcast for the FRCA Primary exam. Our goal is to fill your brain with all the highly useful information you need. You might be in the gym right now, commuting, or ironing your scrubs – no judgement here. Gas, Gas, Gas will prime your brain for the monsoon of knowledge you need to imbibe. Expect facts, concepts, model answers, and the odd tangent. Remember to check out the website – that’s gasgasgas.uk – for show notes with all the detail, links to foundational reference papers, and anything else useful. Buckle up, get ready for your mind to be bent into a new shape, and let’s get on with the show.
[00:56–01:25] It is Tuesday the 17th of February. Today we are covering cisatracurium. We are going to cover its pharmacology, pharmacokinetics, and pharmacodynamics; its similarities with atracurium and other NMBDs, as well as its differences; the ways and means of potentiating and inhibiting the effects of NMBDs in a patient; and we will remind ourselves of the structure and function of the neuromuscular junction, because you certainly might be asked to draw that in an exam. Without further waffle, let’s get on with the show.
Classification, Formulation, and Development
Key points:
- Cisatracurium besylate – also marketed as Nimbex (original) or Cisblock
- One of 10 diastereoisomers of atracurium, isolated in 1989 by Burroughs Wellcome (now GSK)
- Classified as a bis-benzylisoquinolinium ester; non-depolarising competitive NMBD
- 5 mg/ml solution, pH 3.2–3.65, refrigerate; incompatible with propofol, barbiturates, and ketorolac
- Molecular weight 929 g/mol – same as atracurium
[01:26–02:25] Right, we are going to pile straight into cisatracurium pharmacology. If you were to look at it on an ampoule, it would be described as cisatracurium besylate. You might see the brand name Cisblock – which is, obviously, very exceedingly novel – but also Nimbex was its original brand name, apparently short for ‘excellent neuromuscular blocker’. It is classed as a bis-benzylisoquinolinium ester. It is one of the 10 diastereoisomers of atracurium, and part of the group of non-depolarising competitive neuromuscular blocking drugs.
[02:26–03:55] In case we are not quite sure what a diastereoisomer actually means – well, we just say it, don’t we? A diastereoisomer refers to an isomer that has multiple chiral centres, leading to situations where some parts of the molecule appear as a mirror image of parts of another molecule, whilst other parts do not. Atracurium has four chiral centres. Cisatracurium was originally isolated and synthesised from atracurium in 1989 by Burroughs Wellcome – now owned by GlaxoSmithKline. It is presented as a clear, colourless or sometimes pale yellow aqueous solution in 5, 10, and 20 ml vials, at a concentration of 5 mg/ml. It must be kept in the fridge and is quite acidic with a pH of 3.2–3.65. This acidity means it does not play terribly well when mixed with barbiturates, propofol, or ketorolac. Its molecular weight is exactly the same as atracurium’s at 929 g/mol – a chunky molecule.
[03:25–04:24] Why did they really bother to develop cisatracurium? Well, think of it like levobupivacaine: we know that is generally a bit less cardiotoxic than standard bupivacaine. When it comes to cisatracurium, we know that if a patient is particularly prone to histamine release, you would avoid atracurium – and certainly avoid mivacurium – because triggering histamine release can lead to hypotension, bronchospasm, flushing, and so on. Cisatracurium, even in very large doses – up to eight times the ED95 – causes very little histamine release. This is its chief marketable feature. Although, if you look in most anaesthetic fridges, the odds are you probably will not find cisatracurium. I have seen it once in a teaching hospital and thought, ‘Oh, cisatracurium!’ But alas – when you weigh up all the marketing costs and testing, most of us would simply reach for rocuronium if a patient is particularly histamine-prone.
Pharmacodynamics, Dosing, and Side Effect Profile
Key points:
- Mechanism: competitive non-depolarising NMBD – antagonises acetylcholine at nicotinic receptors on the postsynaptic skeletal NMJ
- 3× more potent than atracurium; intubating dose 0.15 mg/kg (2–3× ED95); ED95 = 0.05 mg/kg
- Maintenance: one-fifth of intubating dose by infusion
- Stable cardiovascular profile; bradycardia ~0.5%; bronchospasm ~0.2%; rash ~0.1%; flushing ~0.2%
[04:24–05:22] What about the pharmacodynamics and side effect profile of cisatracurium? Its mechanism of action: competitive non-depolarising neuromuscular blocker, antagonising acetylcholine at the nicotinic receptor on the postsynaptic side of the skeletal NMJ. That is its chief site of action, noting that it would also work on nicotinic acetylcholine receptors found on the presynaptic side of the NMJ. In terms of dosing, it is quite a small dose, because it is three times as potent as atracurium. Whereas atracurium requires approximately 0.5 mg/kg for a reliable, not-too-long-to-wait dose, with cisatracurium you need 0.15 mg/kg. That is a 2–3× ED95 dose.
[05:23–05:52] You can infuse it, and it is recommended to give one-fifth of that dose as a maintenance infusion to tick things along during your case. In terms of onset, naturally if you give more, it works quicker; if you give less, it takes longer. That is why it is sometimes tempting to give just a little bit of rocuronium or atracurium – accepting that you will be ventilating the patient for a little longer – but it will wear off more quickly, which is useful for a high-turnover list. The ED95 for cisatracurium is 0.05 mg/kg to achieve 95% twitch suppression.
[05:53–06:55] Side effect profile. As I have alluded to, it is a very stable drug. Mean arterial pressure and heart rate remain stable with rapid, large boluses of up to eight times the ED95. There is an incidence of approximately 1 in 200 patients developing a bradycardia. It is quite a forgiving drug if you get your dosing slightly wrong – it is probably better to think in micrograms rather than fractions of a milligram. You see a 0.2% bronchospasm rate, a 1 in 1000 rash rate (0.1%), and a 2 in 1000 flushing rate (0.2%). So a very safe and stable drug – not at all like suxamethonium with its hyperkalaemia, sux apnoea, anaphylaxis risk, malignant hyperthermia risk, bronchospasm risk, and bradycardia.
Pharmacokinetics
Key points:
- Volume of distribution: 0.1–0.16 L/kg (small, like other NMBDs)
- Crosses the placenta – consider timing relative to delivery in GA caesarean section
- Primary metabolism: Hofmann elimination (spontaneous, pH- and temperature-dependent)
- Secondary metabolism: hydrolysis by non-specific plasma esterases
- Metabolite: laudanosine – hepatically metabolised, renally cleared; implicated in lowering seizure threshold at very high concentrations
- Cisatracurium produces significantly less laudanosine than atracurium (~20–38 vs ~190–200 micrograms/litre)
- Half-life: 22–35 min; clearance: 4.6–25.7 ml/kg/min; ~4% faecal; ~10–15% excreted unchanged in urine
[06:55–07:23] What about the pharmacokinetics of cisatracurium? Remember, these align pretty consistently with atracurium, because – hey presto – it is part of that bundle of isomers found in an atracurium ampoule. As with most neuromuscular blocking agents, the volume of distribution is small: 0.1–0.16 L/kg. Of note, like atracurium, it crosses the placenta.
[07:23–07:53] So if you have had to perform a GA caesarean section and ventilation is not quite right, try to get the tracheal tube in after the baby is delivered. Otherwise the paediatricians are going to be scratching their heads wondering why the neonate is not breathing, even though it is rather obvious they have had a general anaesthetic.
[07:54–08:52] Now for metabolism. The primary means of metabolism of cisatracurium is Hofmann elimination – where the link between a quaternary nitrogen and the central chain is cleaved. This is a spontaneous, temperature- and pH-dependent process requiring no enzymatic contribution. Its secondary mode of metabolism is hydrolysis by non-specific plasma esterases – esterases floating around in the plasma that, on encountering a cisatracurium molecule, simply get on with the job. Remember: it is chiefly Hofmann elimination. What byproducts are there? Just as with atracurium, the main metabolite is laudanosine. Laudanosine is metabolised by the liver, and that metabolite is cleared renally. It is implicated in lowering the seizure threshold.
[08:53–09:22] Elevated laudanosine levels have not been shown to cause seizures in humans, but there is evidence that plasma concentrations above approximately 17,000 micrograms/ml cause seizures in animals. Now, here is another marketing angle that the pharmaceutical companies were keen to push. Naturally, they ran the studies comparing laudanosine generation with a dose of cisatracurium versus atracurium. And indeed, cisatracurium yields significantly less laudanosine than atracurium – by roughly an order of magnitude: 190–200 micrograms/litre for atracurium versus only 20–38 micrograms/litre for cisatracurium.
[09:23–09:52] So if you have a patient with a severely impaired liver who needs prolonged paralysis on the intensive care unit and you are worried about laudanosine accumulation – particularly if they have been having seizures – then you might think about cisatracurium. But that is a fairly unlikely chain of events, and we do not generally keep patients paralysed for very long. The half-life of cisatracurium is 22–35 minutes. The clearance is 4.6–25.7 ml/kg/min. Approximately 4% is found in faeces, and 10–15% is excreted unchanged in the urine.
Clinical Niche, Relative Potency, and the Bowman Principle
Key points:
- Chief advantages: minimal histamine release, minimal laudanosine production, organ-independent elimination
- Potency relative to other NMBDs: vecuronium > cisatracurium (3×) > atracurium, while rocuronium is 1.3× less potent than cisatracurium
- Bowman’s principle: potency is inversely related to speed of onset – lower potency (e.g., rocuronium) → faster onset
- Cisatracurium not widely stocked in UK clinical practice; rocuronium preferred
[09:53–10:52] So we have mentioned a few reasons why you might be tempted to pick cisatracurium. We know we like it because it does not cause histamine release. We appreciate its minimal laudanosine metabolite status. And naturally, these benzylisoquinolinium compounds are less dependent on the liver and kidney for their effect to wear off – and that is what we are really interested in. You do not really mind if a patient is full of a drug, as long as that drug is not doing anything. But did it catch on? Not really in the UK by the look of things, especially not now. Cisblock or Nimbex perhaps used more elsewhere where budgets allow.
[10:53–11:22] Where would you put cisatracurium in your mental model relative to other NMBDs? In terms of potency: cisatracurium is 3 times the potency of atracurium. Vecuronium is 4–5 times the potency of cisatracurium, whereas rocuronium is 1.3 times less potent than cisatracurium. We know clinically that rocuronium is less potent, requiring larger doses. However, this is clinically useful: you achieve a greater concentration gradient between plasma and the effect site, delivering more drug to that site more rapidly. This is Bowman’s principle – potency is inversely related to onset. The lower the potency, the faster the onset. Bowman’s principle: one to remember.
Sponsor: Teach Me Anaesthetics
[11:22–12:57] Time for a brief mention of the sponsor of Gas, Gas, Gas: Teach Me Anaesthetics. These folks are behind an excellent single best answer question resource. They have over 1,100 questions written specifically for the FRCA Primary exam. When I was studying, this resource did not exist – there were other packages online that cost considerably more and did not really reflect the exam content: I came across rehashed MRCP questions, which was not particularly inspiring. These SBA questions have been built from the ground up. They have not sniffed an MCQ in a past life. They are based on the anaesthetic science you need to know, and there are explainers with every question so it builds your knowledge as you work through them. You can do a random run of 1,100+ questions, split by subject area, or return to the ones you got wrong. Very malleable. If you are thinking about which question resource to use, you should not really need to look further than Teach Me Anaesthetics. Links from Gas, Gas, Gas are affiliate links – so if you sign up through one, you are helping support the podcast. And if you need any more convincing: it’s your number-one, tell-your-nan-at-Sunday-lunch podcast. You know you want to.
Neuromuscular Junction – Structure and Function
Key points:
- Presynaptic terminal bouton contains ACh-filled vesicles; postsynaptic membrane is deeply furrowed (~10,000 nAChRs per µm²)
- Synaptic cleft is ~70 nm wide
- Action potential → voltage-gated Ca²⁺ channels open → SNARE protein activation → ACh exocytosis
- Nicotinic ACh receptors (nAChRs): pentameric, ligand-gated, cation channels (primarily Na⁺)
- Na⁺ influx → depolarisation → ryanodine receptor activation → Ca²⁺ release from SR → excitation-contraction coupling
- Presynaptic nAChRs provide positive feedback to increase ACh vesicle mobilisation – blockade of these causes ‘fade’
[13:01–13:21] In the rocuronium episode, we spoke in relative detail about the neuromuscular junction from a physiological perspective. The concept we are going to explore in the latter half of this episode is considering how NMBDs are potentiated, and looking at patients who are either insensitive or sensitive to NMBDs. Let me remind you about the NMJ – its structure and function – before we get into potentiation and sensitivity.
[13:22–14:20] Think about the structure first. You have a presynaptic membrane where you will find the terminal bouton of your motor nerve, full of acetylcholine-filled vesicles ready to exocytose into the synaptic cleft – the gap between the excitable neuronal membrane and the excitable postsynaptic muscular membrane. That gap is only 70 nanometres wide. The postsynaptic membrane is deeply furrowed to increase surface area and achieve a high density of nicotinic acetylcholine receptors: 10,000 per micrometre squared.
[14:21–15:21] Functionally: an action potential travels down that motor axon at 100–120 m/s if it is an alpha motor neuron. At the presynaptic membrane, voltage-gated calcium channels open in response to membrane depolarisation. The calcium entering the cell activates SNARE proteins, triggering exocytosis of acetylcholine vesicles into the synaptic cleft. More acetylcholine is released than is strictly necessary – there is a substantial safety factor. Within the cleft, acetylcholinesterases rapidly hydrolyse acetylcholine. Some, however, crosses to activate the nicotinic acetylcholine receptors on the postsynaptic membrane.
[15:21–15:50] Do not confuse nicotinic and muscarinic acetylcholine receptors. Nicotinic receptors are the ones at the muscle end of the NMJ – remember: Nicotinic = Neuromuscular. Muscarinic receptors are found in the parasympathetic nervous system and are G protein-coupled receptors. Nicotinic receptors are pentameric ligand-gated cation channels that conduct sodium ions into the cell when activated by acetylcholine. This sodium influx depolarises the postsynaptic membrane.
[15:50–16:19] Within the deep clefts, voltage-gated sodium channels continue and propagate this depolarisation, allowing large amounts of sodium into the muscle cells. This sodium influx then triggers ryanodine receptors on the sarcoplasmic reticulum, releasing stored calcium to initiate excitation-contraction coupling.
[16:20–16:49] There are also presynaptic nicotinic acetylcholine receptors that are activated by the released acetylcholine. This triggers mechanisms within the terminal nerve ending to mobilise more acetylcholine vesicles towards the membrane – a positive feedback loop enabling the nerve to respond to repeated signals to contract. So that was a whistle-stop tour of the neuromuscular junction.
Single Best Answer Question: Neuromuscular Junction
[16:50–18:49]
Mechanisms of Skeletal Muscle Paralysis Beyond NMBDs
Key points:
- Presynaptic synthesis blockade: hemicholinium blocks ACh synthesis → slow paralysis
- Depletion of ACh stores: tetanus toxin
- Blockade of ACh release: gentamicin, magnesium
- Volatile anaesthetics: bind to a site on the nAChR, contributing to NMJ blockade (potentiated at higher MACs)
- MAC 1.7 isoflurane ≈ MAC 1.3 sevoflurane for NMJ synergy; at MAC ~2 sevoflurane, intubation possible without NMBD
- Lithium: mimics Na⁺, accumulates intracellularly, causes partial depolarisation and potentiation of NMBDs
[19:21–20:20] Now let’s think about ways you can interfere with the neuromuscular junction and stop it working – because they may well ask you in the exam to describe how skeletal muscle can be paralysed. It truly depends on the site of action of the agent. You would be remiss to simply say ‘depolarising and non-depolarising NMBDs’, because you have only really addressed the nicotinic receptor side of things. If you stop that nerve ending from synthesising acetylcholine, or make it synthesise slightly defective acetylcholine, that nerve ending simply will not work.
[19:21–19:50] There is a compound called haemicholinium, which you can give to rats (probably not humans), that blocks acetylcholine synthesis and will subsequently cause paralysis, slowly but surely. You could deplete your stores of acetylcholine – and that is what tetanus toxin does. You could prevent acetylcholine from being released from the nerve ending: gentamicin and magnesium are involved here.
[20:21–21:13] You should also consider other drugs that act upon the nicotinic acetylcholine receptor. Chiefly, volatile anaesthetics can bind to a site on the nicotinic acetylcholine receptor and cause a degree of blockade. You have probably seen it yourself: if someone has a MAC of 1.1 and you do a train-of-four, there is quite significant fade. Then you turn off the gas and allow the MAC to fall to around 0.6 – not wanting them to wake up while still paralysed – and when you recheck the train-of-four, the amplitudes are better. Some of that improvement is simply time passing and the NMBD abating, but you have also eliminated a significant amount of volatile agent. In terms of comparative potencies for this NMJ synergy: MAC 1.7 of isoflurane is equivalent to MAC 1.3 of sevoflurane. It is quite synergistic – which is why, at a MAC of around 2 with sevoflurane, you could theoretically intubate someone, although they do tend to cough and buck and it is not particularly pleasant.
[21:44–22:13] Lithium also potentiates NMBDs. Lithium mimics sodium and does indeed pass through activated sodium channels into excitable tissues. However, cells are not well equipped to actively extrude lithium – there is no evolutionary pressure to do so. Lithium therefore accumulates intracellularly. High intracellular lithium concentrations displace intracellular potassium, leading to partial depolarisation of these membranes. So a patient with a moderate lithium level on board might be a little more sensitive to NMBDs, and a patient with a significantly elevated lithium level may be rather floppy.
Patients Insensitive to Neuromuscular Blocking Drugs
Key points:
- Burns patients: upregulation of extrajunctional nAChRs → relative resistance to non-depolarising NMBDs (but DO NOT give suxamethonium after 48 hours due to hyperkalaemia risk)
- Evidence for degree of resistance in burns is mixed – rocuronium distributes throughout the body and may reach extrajunctional receptors anyway
- Enzyme-inducing drugs (phenytoin, phenobarbital): speed hepatic clearance of rocuronium/vecuronium; also increase alpha-1 acid glycoprotein → higher protein-bound fraction of atracurium → smaller free fraction → prolonged onset
- Chronic dantrolene (ryanodine receptor antagonist): may upregulate ryanodine receptors → partial resistance
[22:14–23:13] So now, thinking a little more broadly – how should you approach this in an exam? What about patients who are insensitive to NMBDs – that is, they might need a larger dose or the drugs wear off more quickly? You can break this down into other drugs that relax skeletal muscle and other drugs that alter the pharmacokinetics of NMBDs, as well as disease processes. Consider patient insensitivities and sensitivities as sub-classifications.
[22:44–23:48] One drug that comes to mind is dantrolene – an intracellular skeletal muscle relaxant that antagonises the ryanodine receptor. If no calcium gets out of the sarcoplasmic reticulum, excitation-contraction coupling cannot occur. Are you going to mix dantrolene – which is notoriously tricky to prepare – just to look clever with your anaesthetic? No. But we do occasionally encounter patients taking oral dantrolene for muscle spasticity, and it is worth being thoughtful. If they have just started dantrolene, you might expect them to be somewhat floppy. If they have been on it for years, there is a reasonable argument that they may have upregulated their ryanodine receptors to compensate for chronic exposure – and they might actually be relatively more resistant.
[24:13–25:01] In fact, there is a plausible argument that chronic exposure to any neuromuscular blocking drug would lead to progressive upregulation of nicotinic acetylcholine receptors, and relative resistance to that drug. The same principle applies to burns patients: there is a well-established phenomenon of extrajunctional acetylcholine receptor upregulation in the weeks and months following a significant burn. This confers relative resistance to non-depolarising NMBDs. However, whether this is clinically significant is debated. Rocuronium distributes throughout the body – it does not simply head straight to the NMJ like a Monopoly player passing Go. It bathes all the surrounding muscle tissue. So the concentration at the NMJ is probably much the same regardless. Those extrajunctional receptors that are blocked are not normally involved in coordinated muscle contraction anyway, as they are not innervated. However, do not give suxamethonium to burns patients after 48 hours: triggering all those extrajunctional receptors would cause a massive efflux of potassium and potentially fatal hyperkalaemia.
[25:43–26:42] What about pharmacokinetic interactions with other drugs? The first thing that should come to mind is enzyme-inducing drugs – drugs that induce hepatic enzymes, thereby speeding up the metabolism of agents cleared by the liver. Think rocuronium and vecuronium. But also consider drugs that increase plasma protein concentrations: phenytoin and phenobarbital increase the production of alpha-1 acid glycoprotein. Atracurium has a relative affinity for alpha-1 acid glycoprotein, so you will end up with a smaller unbound fraction of your dose, and potentially a prolonged onset time.
[26:43–27:12] As an interesting aside: I am not entirely sure whether chronic phenytoin or phenobarbital use is particularly protective against local anaesthetic systemic toxicity (LAST). Local anaesthetic also binds to alpha-1 acid glycoprotein. However, phenytoin is itself a sodium channel blocker. If you have local anaesthetic floating around blocking sodium channels and causing mischief, and you also have phenytoin blocking sodium channels, it is probably not going to help your LAST. And if someone is having seizures due to LAST, you would probably not reach for phenytoin anyway – the Quick Reference Handbook recommends benzodiazepines first for seizures, and propofol second.
[27:13–27:42] It is also worth noting that anti-epileptics have a membrane-stabilising effect – they stabilise neuronal membranes in the brain to reduce seizure likelihood, but they also have effects on other excitable membranes in the body. That is why phenytoin can cause ventricular arrhythmias and requires ECG monitoring during IV loading.
Patients Sensitive to Neuromuscular Blocking Drugs
Key points:
- Autoimmune conditions: polymyositis, dermatomyositis, lupus
- Neuromuscular disease: myasthenia gravis, Guillain-Barré syndrome, motor neuron disease, myotonic and muscular dystrophies
- Drug interactions: aminoglycosides (e.g., gentamicin) potentiate NMBDs
- Management: if paralysis required, use reduced-dose rocuronium + appropriate-dose sugammadex
- If intubation only (no paralysis needed): remifentanil-facilitated intubation is a safe alternative
[27:43–28:42] What about patients who are more sensitive to NMBDs? Certain conditions and drugs increase sensitivity. You can break these conditions into those with an autoimmune basis impairing muscle function, and those primarily affecting neuromuscular function. On the neuromuscular side: motor neuron disease, myasthenia gravis, Guillain-Barré syndrome, myotonic and muscular dystrophies. On the autoimmune side: polymyositis, dermatomyositis, and lupus. Drug-wise: you might think carefully about whether you want to give gentamicin to a patient with urosepsis and myasthenia gravis. A one-off dose is probably fine, but if you are on a 7 mg/kg regimen with peak and trough monitoring, you might find yourself with a more myasthenic patient than you started with. Admittedly, a myasthenic patient with an active infection might deteriorate anyway purely due to the infection.
[28:43–29:14] It is worth being thoughtful and considerate about your drug choices for neuromuscular blockade in these patients. If you need to paralyse them, reduced-dose rocuronium with appropriately dosed sugammadex is considered the appropriate strategy. However, if you just need to get a tube in without necessarily needing ongoing paralysis, remifentanil-facilitated intubation is our friend – we know we can achieve intubating conditions in a deeply anaesthetised patient with an appropriate remifentanil dose.
Episode Wrap-Up and What’s Coming Next
[29:15–29:41] Thank you everyone for listening. I quite enjoyed digging into the detail with cisatracurium, but I think I enjoyed even more thinking about the interesting ways you can inhibit the function of the neuromuscular junction. I do wonder – when we get to an episode on dantrolene, which we will – whether there is an IV dantrolene dose that could facilitate intubation. Perhaps I will look into that, just for a laugh. In two weeks’ time we are going to cover either vecuronium, pancuronium, or both, depending on how much material there is. Then we will cover sugammadex and perhaps an episode on the more historic neuromuscular blocking agents – D-tubocurarine and gallamine – just for the sake of completeness.
[29:42–30:12] Happy Friday, and I hope you have a lovely weekend. Ahoy, Team Anaesthesia! You have survived yet another episode of Gas, Gas, Gas. If you found it useful – or harrowingly awful – please like and subscribe, drop us a rating or 12, and follow on whichever podcast platform you use. Please leave a comment or send an email if you think I need to sort something out. There are various ways to support the costs of Gas, Gas, Gas, from buying me a coffee to using an affiliate link through to Teach Me Anaesthetics. Those links are on the website and in the show notes. Check out gasgasgas.uk for show notes, diagrams, details, and references.
[30:42–30:53] Take it day by day. Don’t overcook yourself. Don’t freak out. And keep studying.
Leave a Reply