Anticholinesterases For The Anaesthetically Adept
19 April 2026
Contents
Introduction
Anticholinesterases are drugs that inhibit the enzyme acetylcholinesterase, preventing ACh breakdown and amplifying cholinergic transmission at both nicotinic and muscarinic receptors.
Anticholinesterases. You think you know what you’re getting into: neostigmine, reversal, done.
Then you realise it can go considerably further than that. Nerve agents. Myasthenia gravis. Alzheimer’s disease + classification based on where exactly a drug binds to an enzyme that has more than one active site.
This episode works through all of this. We start with the classification of anticholinesterases by mechanism, cover the pharmacology of neostigmine in some detail, deal with the muscarinic side effects and why glycopyrronium tags along, and then zoom out to look at acetylcholine itself: where it comes from, how it’s stored and released, and how the nicotinic acetylcholine receptor actually works. There’s a Teach Me Anaesthetics question in the middle as well.
AntiCholinesterase Pharmacology
Anticholinesterases inhibit acetylcholinesterase, increasing ACh availability at nicotinic and muscarinic receptors; they are classified by binding site — edrophonium (anionic), carbamates such as neostigmine (anionic and esteratic), and organophosphates (irreversible esteratic phosphorylation).
Physico-Chemical properties
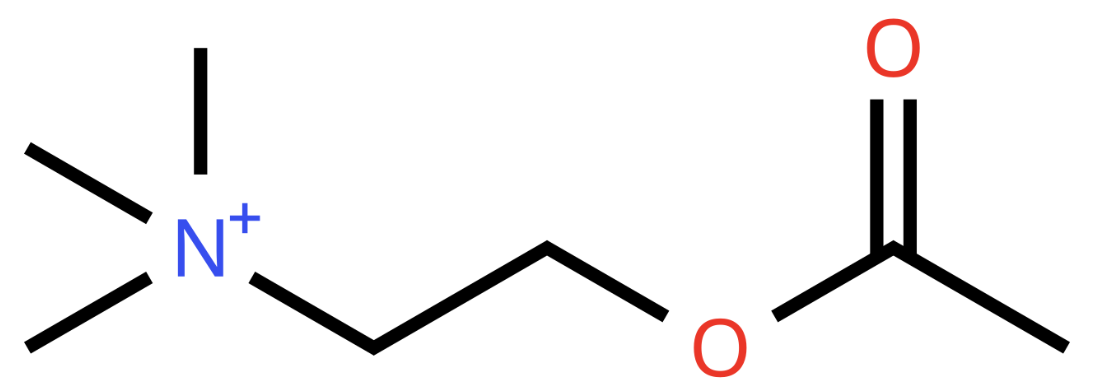
| Names | Edrophonium : Neostigmine : Pyridostigmine : Organophosphates (And Donepezil and Rivastigmine) Neostigmine is a quaternary amine Ester of alkyl carbamic acid (quaternary ie can’t cross into the CNS) |
| Class | Anti-Cholinesterases which are parasympathomimetics Work by stopping the enzyme acetylcholinesterase from being able to bind ACh This leads to more ACh Available for Muscarinic and Nicotinic Acetyl choline receptors throughout the body |
| Chemical Make Up | Edrophonium: Quaternary ammonium compound, phenol based Fancies the look of the ANIONIC site. Neostigmine/ Pyridostigmine / Physostigmine: Carbamate esters (which carbymalate) Binding to Both ANIONIC and ESTERATIC sites! Organophosphates (insecticides + Sarin/ Novichok) These Phosphorylate the esteratic site irreversibly binding to it. (you need to block the effects of ACh to have any chance if the exposure is significant (atropine) and then Pralidoxime promotes hydrolysis of the phosphate of the organophosphate molecules, treating the issue. (the british armed forces, in high risk situation take pyridostigmine pre-treatment (NAPS) to reduce the chance of such poisoning) |
| History | |
| Uses | Myasthenia Gravis NMB reversal Ileus (some flatulent reading here) |
| Colour/Appearance | As a clear colourless, solution for injection. 2.5 mg/ml |
| Molecular weight | 223.6 g/mol |
Acetylcholinesterase, found deep down in those junctional clefts of the NMJ (the nicotinic receptors sit on the tops of those clefts)
Has an Esteratic and a Anionic Binding site.
Anionic binds with quaternary ammonium compounds
Esteratic likes the ester group of ACh
It Acetylates Acetylcholine, which gets hydrolysed producing acetic acid (vinegar!)
Anticholinesterases Pharmacodynamics & Side Effects
| Mechanism of Action | Remember that [Rocuronium](/episode/rocuronium/) is in competition with acetylcholine (ACh) for the nicotinic receptor in the NMJ, the Ach triggers the Nicotinic receptor, rocuronium does not when it binds. the balance between these two molecules is what dictates paralysis, more rocuronium, more paralysis. So if we could bolster the acetylcholines ranks int he NMJ it will out compete and shift away from paralysis. Do remember tho, this system has a wide error margin, you need quite a lot of receptor occupancy to achive paralysis, and once we start seeing twitches there are not many receptors available for the ACH at that point in time, when the NMJ is ‘at rest’ with only a gentle rocking of the boat occuring as rocuronium is slowly metabolised from the plasma, and it slowly seeps out of the NMJ. We throw an anticholinesterase in the mix, and now were swaying across the north sea (with perhaps some vomiting/PONV) and the rocuronium is now being displaced given the higher fraction of ACh in the NMJ relative to the rocuronium concentration. Only 1 twitch seen = 95% or so receptor occupancy 3 twitches seen (T4 lost) = 90% occupancy 4 twitches = 70% occupancy But if you were to somehow fill a patient up to the eyeballs with an anti-cholinesterase, you would in fact paralyse them, as the excess of ACH leads to continuing depolarisation of the muscle! |
| Chief Effect / Actions | Far from magical reversal of competitive non-depolarising neuromuscular blocking drugs. Onset of Neostigmine = 1 minute, lasting for 40-60 minutes Peak Effect by 7-11 minutes |
| Dose | 50-70 mcg / kg iv Adults: 2.5-5mg (but requires anti-muscarinic agent alongside) Accidental bolus of unopposed neostigmine can lead to asystole. Slow IV administration can avoid this… and is sometimes used when ICU really want someone to poop. (Patients were randomly assigned to receive 2.0 mg of neostigmine intravenously over a period of three to five minutes or identical-appearing saline placebo) |
| Side Effects | Muscarininc/Cholinergic Side effects Lungs : Bronchospasm and increased secretions CVS: Bradycardia, reduced contractility CNS: Miosis, blurred vision, Airway: Hypersalivation GI: PONV + peristalsis ++ Abdo pain / Cramping GU: involuntary micturition |
| Cautions | There is hesitancy in administering neostigmine to patients who have undergone primary anastomosis, due to its colonic peristaltic action. This is opposed by glycopyrollate, but this problem is somewhat in the bin (if it did exist) courtesy of [sugammadex](/episode/sugammadex/). |
Neostigmine Pharmacokinetics
| Absorption | 1-2% oral Bioavailability (hence why not used with myasthenia) |
| Distribution | Volume of distribution: 0.4-1 L/KG No plasma protein binding |
| Metabolism | Plasma esterases > quaternary alcohol – some hepatic metabolism |
| Elimination | Half-life = 50-90mins 50-67% in urine Clearance 5.7 – 11.1 ml/kg/min |
Acetylcholine in Anaesthesia
Acetylcholine is synthesised from acetyl-CoA and choline (via choline acetyltransferase), stored in presynaptic vesicles, and released by calcium-triggered SNARE-protein exocytosis; it acts on pentameric nicotinic receptors (2α, β, ε, δ subunits) at the NMJ.
How is acetylcholine Synthesised
We need Acetyl-Coenzye A (krebs cycle) and some choline (diet), Combined by Choline Acetyl Transferase. (in your nerve endings)
Stored in?
Synaptic Vesicles that are present right at the end of the presynaptic membrane, with stores held back in reserve in the pre-synaptic bouton
Released how?
action potential hitting the end of an axon, increases calcium concentration in the synaptic bouton, triggering a chain of events that cause vesicles to merge with and empty there contents across the membrane into the synaptic cleft. Mediated by a gaggle of proteins whose umbrella terms is SNARE proteins. Check out a painfully deep dive on the current (but still imperfect) understanding of these proteins Remember Tetanotoxin and Botulinum toxin both mess with SNARE (in different ways)
Acts on?
Nicotinic Acetylcholine receptors, which are pentameric ion channels (5 sub-units – two Alpha, single Beta, epsilon and a delta) remember there is a fetal nicotinic Ach receptor also, Check out the suxamethonium post for a lovely drawing of the nicotinic ach receptor channel complex
Na / K / Ca will cross this channel
Summary
That’s anticholinesterases done. Neostigmine, pyridostigmine, edrophonium, organophosphates, the anionic and esteratic sites, SNARE proteins, the fetal nicotinic receptor. It’s more ground than it looks on paper, and if you’ve made it to the end, you’re in good shape.
Show notes are at gasgasgas.uk, with references and anything else worth reading on this one. If the episode was useful, a rating or a follow on your platform of choice goes a long way. Take it day by day, keep studying, and we’ll see you on the next one.
References
- Appiah-Ankam J, Hunter JM. Pharmacology of neuromuscular blocking drugs. BJA Education 2004;4(5):164–8. https://www.bjaed.org/article/S1743-1816(17)30598-X/fulltext
- https://pmc.ncbi.nlm.nih.gov/articles/PMC9490555/
- Peck & Hill, Pharmacology for Anaesthesia and Intensive Care, 4th ed, (chapters on NMBS)
Thanks for listening. Take it day by day, don't overcook yourself — keep studying.
Transcript
20 min listenRead the full transcript
Anticholinesterases — Gas Gas Gas Podcast
Introduction
00:00 – 01:00
Section Summary
- Standard Gas Gas Gas intro and housekeeping
- Episode framing: anticholinesterases are more complex than they first appear
- Show notes and references at gasgasgas.uk
Please listen carefully.
Hello, Team Anaesthesia. Welcome to Gas Gas Gas — the best anaesthetic science podcast for the FRCA Primary exam. Our goal is to fill your brain with all this highly useful information.
Now you might be in the gym right now, commuting, or ironing your scrubs — and there’s no judgement here. Gas Gas Gas will prime your brain for the monsoon of knowledge you need to imbibe. But regardless, the revision is eventually going to end. For now, expect facts, concepts, model answers, and the odd tangent.
Remember to check out the website — that’s gasgasgas.uk. There are show notes there with all the detail, plus links to foundational reference papers and to anything else useful I can find for you.
Anyway, buckle up. Get ready for your mind to be bent into a new shape, and let’s get on with the show.
Framing the Topic: Why Anticholinesterases Matter
01:00 – 03:10
Section Summary
- Anticholinesterases appear broader in the curriculum than many candidates expect
- Clinical uses span myasthenia gravis, Alzheimer’s disease, organophosphate poisoning, paralytic ileus, and neuromuscular blockade reversal
- Neostigmine is the primary focus, given its relevance to anaesthetic practice
- Neostigmine: quaternary amine — does not cross the blood–brain barrier; this is clinically desirable
- Pre-mixed preparation: 2.5 mg neostigmine + 500 micrograms glycopyrronium
Hello everyone, and welcome to yet another episode of Gas Gas Gas. I’m sure you’re deep into the darkest realms of revision fun that you’re currently having — hoping for some brevity and levity, neither of which will be found today, sadly.
Coming across the material you had to know about anticholinesterases for the Primary, I just thought: oh my days. I thought it was one drug that did one little simple thing. Now I feel like I’ve got to learn a load of other things. And it all got very briefly quite emotional.
So we’re going to distil it down so that, hopefully, if someone does throw you that left hook, you’ll at least be able to block it and bat them away.
Whenever you get asked a question in the exam, there’s the really easy answer where you just give them exactly what they want — you answer the question they’ve asked. But oftentimes they’re doing that irritating thing where actually they want you to say more than just: ‘an anticholinesterase — yes, that’s neostigmine, let’s move on.’ When in fact they might want something a little bit more fleshed out.
If you broaden your mind briefly to anticholinesterases in medicine and where you might find them being used: it’s a treatment for myasthenia gravis, and a test to confirm whether a patient has myasthenia. The anticholinesterase donepezil is used in Alzheimer’s disease. Organophosphates — i.e. pesticides — have anticholinesterase activity at quite lethal levels. And then in the anaesthesia and ITU environment, we reach for it sometimes, very rarely, for paralytic ileus — and there’s a link to a New England Journal of Medicine article in the show notes.
And then there is the utilisation of neostigmine in anaesthetic practice for reversing a diminishing competitive non-depolarising neuromuscular blockade.
Why do we use neostigmine? Well, it works for about the right amount of time that we want, has a reasonable onset time when given IV, and it doesn’t cross the blood–brain barrier. It’s a quaternary amine — a bit like glycopyrronium — a quaternary molecule that can’t cross the blood–brain barrier. And that’s because there are some desirable effects that we want as anaesthetists from neostigmine. But unfortunately, as with most drugs — except for the pedestalised, glorious sugammadex — it has side effects. And these are side effects that promote the parasympathetic nervous system’s functions. We’re going to get into those.
If they asked you to classify — and you’re deep into the exam, thinking ‘classify, that’s going to make me cry’ — you need to base it on which site on an acetylcholinesterase molecule the drug works on. And that then raises the horrible question: well, good God, there’s more than one active site on an acetylcholinesterase enzyme. And yes, the answer is there’s an anionic site and an esteratic site.
So just for completeness and clarity, unless otherwise mentioned, I’m just going to talk about neostigmine. Neostigmine is a clear, colourless solution for injection, presented at 2.5 mg per mL, often in a 1 mL ampoule. However, it also comes pre-mixed at 2.5 mg of neostigmine with 500 micrograms of glycopyrronium.
They might jump in there and say, ‘Well, why do they do that?’ Acetylcholine works on nicotinic but also muscarinic receptors. We like the nicotinic effects, but we do not like the muscarinic effects.
Classification of Anticholinesterases by Mechanism
03:57 – 06:20
Section Summary
- Classified by their binding site on acetylcholinesterase
- Anionic site only: edrophonium — short-acting, used in myasthenia testing (Tensilon test)
- Anionic + esteratic (carbamylation): neostigmine, pyridostigmine, physostigmine — reversible, moderate duration
- Esteratic site (phosphorylation): organophosphates — irreversible; includes pesticides and nerve agents (sarin, novichok)
- Pyridostigmine used in myasthenia gravis because of its oral bioavailability; neostigmine has <2% oral bioavailability
- Military pretreatment with pyridostigmine offers partial protection against nerve agent lethality
- Treatment for organophosphate poisoning: atropine (muscarinic blockade) + pralidoxime (reversal of phosphorylated binding)
So first step — physicochemical — classify or cry. What does what?
We’re going to break this down into: drugs that only work on the anionic site; drugs that work on both the anionic and esteratic sites, which carbamylate those sites; and then drugs that phosphorylate the esteratic site.
Edrophonium really fancies the look of that anionic site, but it doesn’t work very long — it just hydroxylates it, then floats off and stops inhibiting that enzyme. So that’s really useful when testing for myasthenia.
And then we’ve got our more common neostigmine, but also pyridostigmine and physostigmine. Pyridostigmine is used to treat myasthenia. These are carbamate esters, which carbamylate and bind to both sites. So they’re quite good at causing inhibition for a reasonable period of time.
Why don’t we give myasthenics oral neostigmine? The oral bioavailability of neostigmine is abysmal, whereas with pyridostigmine it is not — and that’s why we use pyridostigmine for myasthenia gravis, and neostigmine, with its shorter duration of effect, to reverse neuromuscular blockade.
And then what about those that phosphorylate the esteratic site? That would be organophosphates. These bind pretty much irreversibly to your acetylcholinesterase. You can imagine your mind’s probably thinking that organophosphates are generally bad — that’s like sheep dip and other such things. But it is sheep dip, but also sarin, novichok, and a lot of other nerve agents work on this principle of inhibiting acetylcholinesterase.
This leads to overwhelming muscarinic effects: bradycardia, prolific production of bronchial secretions, bronchospasm — just generally really terribly bad for your health. And can kill you. And does.
The Salisbury poisonings with novichok — the paramedics gave atropine because of bradycardia, and it probably actually saved the life of that police officer, because atropine is an emergency treatment for nerve agent poisonings such as this.
In the army, you take pretreatment in high-risk environments — pyridostigmine three times a day — in order to block the potential for a nerve agent to be lethal. And then you carry an auto-injector containing atropine and a few other molecules, presumably including pralidoxime, which is something that will reverse the effect of that phosphorylated binding to the esteratic site of your acetylcholinesterase.
Sponsor: Teach Me Anaesthetics
06:21 – 07:50
Anyhow, time for a brief mention about the sponsors of Gas Gas Gas — Teach Me Anaesthetics. These guys are behind an excellent single best answer question resource. I have taken the joyful, challenging, and intermittently bruising journey through their 1,100-plus questions, which they have written specifically for the FRCA Primary exam. I secretly loved it, and there are plenty of questions to test your knowledge.
When I was studying for the FRCA Primary, it did not exist. But there were other packages online. These cost a lot more and didn’t really seem to reflect the content of the exam. Whilst doing them, I came across rehashed MRCP questions, which was not terribly inspiring and left me pretty cheesed off — whereas these single best answer questions have been built from the ground up. They have not sniffed an MCQ in a past life. They are based on the anaesthetic science you need to know, and there are explainers with all the questions so it builds your knowledge as you work through them.
There are a multitude of ways you can study: you could do a random battle with 1,100-plus questions, or split them into subject area. You can go back and redo the ones you’ve got wrong. It’s very malleable. So if you’ve been thinking about which question resource you might want to get your hands on for the exam, you shouldn’t really look any further than Teach Me Anaesthetics.
If you reckon it’s for you, all links to Teach Me Anaesthetics from Gas Gas Gas are affiliate links — so if you’re signing up through one of those, you’re helping to support Gas Gas Gas. You’ll not only be supporting your exam preparation, but you’ll be supporting your 100% favourite ‘tell your nan over Sunday lunch’ podcast, which is of course Gas Gas Gas.
Mechanism of Action in Anaesthetic Practice
07:50 – 10:00
Section Summary
- Neostigmine and rocuronium compete for the nicotinic acetylcholine receptor (N2 receptor) at the neuromuscular junction
- Four twitches can still be present with up to 70% receptor occupancy — the neuromuscular junction has a significant safety margin
- Neostigmine has a ceiling of effect — cannot reverse a dense RSI-dose rocuronium block
- Overdose of neostigmine in the absence of neuromuscular blocking agent can paradoxically cause paralysis (depolarising block from acetylcholine excess)
- This is also a mechanism of organophosphate lethality
Coming back to the slightly more anaesthetically flavoured moment: we need to remember that rocuronium, our quintessential neuromuscular blocking drug, is a competitive non-depolarising muscle relaxant, and it is competing with acetylcholine. They’re in the ring, they’re duking it out — hopefully both of them have boxing gloves on — competing to bind to that nicotinic acetylcholine receptor, sometimes written as the N2 receptor.
The balance between these molecules in the neuromuscular junction dictates the level of paralysis in the patient. More rocuronium, more paralysis; less rocuronium, less paralysis. Remember that you can have four twitches in a patient but be seeing 70% receptor occupancy of the nicotinic receptors. There is a significant margin of error in that system — because presumably you want to be able to ambulate and breathe, and if you were to have just a whiff of rocuronium — or lick a couple of frogs in some jungle — you wouldn’t want one lick to render you dead. You’d want to be twenty or thirty licks in before you started getting a bit floppy.
But we know from our sugammadex episode that if you were to have an RSI dose of rocuronium and then tried to desperately reverse it with neostigmine, it would not go your way. And that’s simply because there’s a ceiling of effect. You can only get so much acetylcholine to the neuromuscular junction. It is being out-competed by the rocuronium in those early stages of the problem of having given rocuronium that you can’t reverse.
So neostigmine’s mechanism of action is to facilitate acetylcholine competition in the neuromuscular junction.
Now, if you were to give loads of neostigmine carefully to a patient who didn’t have rocuronium on board, eventually you would flood that neuromuscular junction with acetylcholine and you could paralyse them — because you’ve depolarised their muscles. There’s so much acetylcholine that the receptor just can’t recover, sodium is hooting in and out of those muscles. And that’s one of the mechanisms by which organophosphates can lead to death — it can just lead to you being paralysed. Lovely.
Pharmacodynamics and Dosing
10:00 – 11:25
Section Summary
- Onset: approximately 1 minute IV; peak effect at 7–11 minutes; duration 40–60 minutes
- Dosing: 5–70 micrograms/kg (0.05–0.07 mg/kg); in adults 2.5 mg is usually adequate, 5 mg if reversal is partial
- Must not be given unopposed — risk of bradyarrhythmia and asystole
- Always co-administer with glycopyrronium (onset ~2 minutes) to mitigate muscarinic side effects
- Timing relative to surgical closure requires clinical judgement
So the onset of neostigmine is about one minute, and it lasts for 40 to 60 minutes. But the peak effect is at 7 to 11 minutes. So you’ve got to time it and weigh it up when you’re using it. You don’t want to be giving it when the patient is awake and all floppy and jerky and unable to use their arms or legs — because that’s quite distressing. But equally you don’t want to give it whilst the surgeon is still sewing up and you’ve only got the barest of four twitches. There is a bit of an art to knowing your surgeon, knowing the intervention, knowing that they don’t actually have to do a stoma — and you’ve been distracted thinking they’ve sewn up the abdomen.
Dosing of neostigmine: it comes in 2.5 mg ampoules. If anyone were to really pin you down, the dose is 5 to 70 micrograms per kilogram, or 0.05 to 0.07 mg/kg. Generally speaking in adult practice, 2.5 mg is fine. And if you’re not quite sure and only the fourth twitch has just reappeared, maybe you’ll give 5 mg.
Should you give it unopposed? No. An accidental bolus of unopposed neostigmine can certainly lead to bradyarrhythmias and asystole — which is bad news. That’s why when you’re giving it for paralytic ileus, you give it slowly, and that’s why we give it with glycopyrronium on board. The onset time of both is reasonably similar — glycopyrronium maybe about two minutes to really see something going on — but it’s enough to stop things getting very unpleasant.
Side Effects and the Cholinergic Crisis
11:23 – 12:41
Section Summary
- Side effects are muscarinic (cholinergic) — follow a systems-based approach for exam answers
- CNS/eyes: miosis, blurred vision
- Airway: hypersalivation
- Respiratory: bronchospasm, increased bronchial secretions
- Cardiovascular: bradycardia, reduced contractility
- GI: nausea, increased peristalsis, abdominal cramps — caution with bowel anastomosis
- GU: involuntary micturition
- Neuromuscular: fasciculations, hypotonia, paralysis at excessive doses
- Excess neostigmine or pyridostigmine → cholinergic crisis (same picture as organophosphate toxicity)
- Sugammadex preferred where anastomosis present to avoid GI complications
Side effects are muscarinic — or cholinergic, depending on which terminology you prefer. Breaking that down by system, head to toe:
Central nervous system and eyes: miosis, blurred vision. Airway: hypersalivation. Respiratory: bronchospasm and increased bronchial secretions. Cardiovascular: bradycardia and reduced myocardial contractility. Gastrointestinal: nausea, markedly increased peristalsis, abdominal pain and cramping. Genitourinary: involuntary micturition. You may also see fasciculations and, at excessive doses, hypotonia or frank paralysis.
You could label this picture as a cholinergic crisis. You will see a similar picture developing in a myasthenia gravis patient who has inadvertently been given too much pyridostigmine — they can start developing a cholinergic crisis.
Now, in patients who’ve had a primary bowel anastomosis, do you really want to be causing loads of peristalsis — plus or minus the abdominal pain and cramping? Do you really want a hyperactive segment of bowel right next to an anastomosis? There are some suggestions that you should be tentative about using neostigmine/glycopyrronium reversal in these patients. I don’t know if there’s any robust evidence, but that’s somewhat moot now because we have sugammadex — that beautiful gammacyclodextrin — which solves many of these problems.
Pharmacokinetics of Neostigmine
12:41 – 13:20
Section Summary
- Oral bioavailability: 1–2% — hence IV use only in clinical anaesthesia
- Volume of distribution: 0.4–1 L/kg — predominantly water-soluble; limited plasma protein binding
- Metabolism: plasma esterases → quaternary alcohol; hepatic contribution
- Half-life: 50–90 minutes
- Elimination: 50–67% renal excretion
- Clearance: 5.7–11 mL/kg/min
Pharmacokinetics of neostigmine. This is really quite brief. As I said, 1 to 2% oral bioavailability — that’s why we don’t use it for myasthenia.
Volume of distribution is 0.4 to 1 litre per kilogram. From that we can infer that it is quite water-soluble, probably staying in a similar compartment to rocuronium. It doesn’t bind significantly to plasma proteins. Metabolism is by plasma esterases to a quaternary alcohol, with the liver also playing a part. Half-life is 50 to 90 minutes. 50 to 67% is excreted in the urine, and its clearance is 5.7 to 11 mL/kg/min.
Teach Me Anaesthetics MCQ: Pyridostigmine Mechanism
13:21 – 15:14
Section Summary
- MCQ on mechanism of action of pyridostigmine in a myasthenic patient
- Correct answer: carbamylation of acetylcholinesterase (reversible, unlike organophosphates)
- Key distinction: carbamylation = reversible; phosphorylation = irreversible
- Increased muscarinic acetylcholine → bradycardia, increased GI motility, bronchial secretions → justifies co-administration of glycopyrronium
Acetylcholine: Synthesis, Storage, and Release
15:15 – 17:17
Section Summary
- Acetylcholine synthesised from acetyl-CoA (Krebs cycle) + choline (dietary) by choline acetyltransferase
- Stored in synaptic vesicles at presynaptic terminals throughout the nervous system
- Used in: preganglionic sympathetic, pre- and post-ganglionic parasympathetic, CNS, post-ganglionic sympathetic to sweat glands
- Proximal vesicle reserve mobilised during sustained release — mechanism underlying train-of-four fade
- Release triggered by action potential → calcium influx at terminal bouton → SNARE protein-mediated exocytosis
- Botulinum toxin and tetanus toxin both disrupt SNARE proteins
They could certainly dip into that pathway having spoken so eloquently about neostigmine, pyridostigmine, myasthenia, and organophosphates. So: acetylcholine is derived from two molecules — acetyl-coenzyme A (from the Krebs cycle) and choline (which you obtain from dietary sources). These are combined in nerve endings by choline acetyltransferase, and the resulting acetylcholine is stored in synaptic vesicles.
These vesicles are present right at the end of your presynaptic membranes throughout your nervous system — because acetylcholine is used in your preganglionic sympathetic, in your pre- and post-ganglionic parasympathetic, in your CNS, and post-ganglionic sympathetic when it comes to supplying sweat glands.
It’s stored in synaptic vesicles at the end of these presynaptic membranes, with some stores also held back just proximal to that membrane. This reserve is mobilised when presynaptic acetylcholine receptors are triggered with sustained release — and this is why you get fade on train-of-four monitoring.
How is it released? Your action potential hits the end of the axon — it gets to that terminal bouton — letting calcium into the cell. Calcium triggers a chain of events that leads to vesicles merging with the membrane and emptying their contents into the synaptic cleft. This is mediated by a gaggle of proteins. The umbrella term for them is SNARE proteins. There are quite a few of them, and they anchor the vesicle to the inside of the neuronal membrane in a manner that causes a rapid unzipping effect on exposure to calcium, pulling the vesicle into the membrane and causing exocytosis of the acetylcholine molecules, which then go off trotting across the cleft.
I’ve linked to a really painfully deep dive on the current — but still imperfect — understanding of these proteins in the show notes. Note also that botulinum toxin and tetanus toxin both interfere with SNARE proteins, and that’s how they produce their effects on the neuromuscular system.
The Nicotinic Acetylcholine Receptor
17:17 – 18:45
Section Summary
- Pentameric ligand-gated ion channel: 2α, 1β, 1ε, 1δ subunits (adult); 2α, 1β, 1γ, 1δ subunits (fetal)
- Two binding sites for acetylcholine — one on each α subunit; both must be occupied for channel opening
- Channel conducts Na⁺ and Ca²⁺ in; K⁺ out
- Fetal receptor: γ replaces ε subunit; more sensitive to acetylcholine (compensates for lower ACh synthesis efficiency in fetal nerve terminals)
- BJA article on neuromuscular blocking drugs in infants and children linked in show notes
- Further detail in the suxamethonium episode
You certainly have to know about this receptor, folks. The nicotinic acetylcholine receptor is a pentameric ion channel — pentameric, there are five subunits. There are two alpha subunits, one beta, one epsilon, and one delta. These make up your adult nicotinic acetylcholine receptor.
Now to make your life even harder, there is a fetal nicotinic acetylcholine receptor with different subunits. The difference between the adult and fetal forms is that instead of an epsilon subunit, you have a gamma subunit.
There’s a BJA article that looks at neuromuscular blocking drugs in infants and children that talks a little bit more about the fetal receptor.
The nicotinic acetylcholine receptor has two binding sites for acetylcholine — one on each alpha subunit. When both are occupied, the receptor opens. It is a channel: it allows sodium and calcium into the cell, and potassium out.
The difference with the fetal receptor is that it is a bit more sensitive to acetylcholine. The supposed mechanism for this is that fetal nerve endings are not quite as efficient at making acetylcholine — the turnover, the efficiency, not so good. Therefore, not so much acetylcholine in the neuromuscular junction. So you need a slightly more sensitive receptor to achieve transmission of action potentials and trigger muscle contraction.
There’s a link in the show notes to the suxamethonium episode as well, which has a lovely drawing of a nicotinic acetylcholine receptor if that’s your bag.
Closing Remarks
18:44 – 19:13
And here is the end of the neuromuscular blocking chapter. We have done all the common ones and a few uncommon ones. I’m going to resist the temptation to go down into tubocurarine and gallamine and all that sort of stuff, because we need to be realistic — you are studying for an exam. Maybe that could be a Christmas special. Maybe we can talk about licking frogs next Christmas. Who knows?
No animals are harmed during the making of Gas Gas Gas — but I do sometimes receive harm from an animal, as Frank gets his claws out. Anyhow, hope you’ve had a lovely week. Have a nice weekend. You’ve been listening to Gas Gas Gas. Cheerio and goodbye.
Enjoyed this? Review on Apple Podcasts Rate on Spotify
Support the show Help keep the lights on SBA question bank @ Teach Me Anaesthetics