Ep 10 – Michaelis-Menten Enzyme Kinetics & Law of Mass action
9 November 2024
Contents
Michaelis-Menten enzyme kinetics are a corner stone of hepatic clearance, enzyme activity and directly relate to the concepts of receptor binding and receptor affinity.
It graphically represents the effects of partial vs complete agonism and antagonism.
And this introduction to logarithmic graphs displaying concentration changes in a non-linear manner will ground you for volatile wash in curves, decay kinetics in poly-compartmental models and all that other good stuff.
What is Michaelis-Menten Kinetics?
Michaelis-Menten kinetics are a conceptual framework for understanding enzyme activity in physiological systems. Enzymes are essential for nearly all bodily functions.
Define or Die
- Enzyme: A protein catalyst that facilitates a chemical reaction by lowering the activation energy required, thus often increasing the rate of reaction or enabling a reaction that would not otherwise occur.
- Substrate–Enzyme–Product Reaction: These reactions can be bidirectional. For example, CO₂ + H₂O → Carbonic Anhydrase → H₂CO₃ ↔ HCO₃⁻ + H⁺ (buffered).
Enzyme Inhibitors and Types
Enzyme inhibitors reduce reaction velocity by one of three mechanisms: competitive inhibitors block the active site, non-competitive inhibitors alter enzyme conformation, and allosteric modulators require a cofactor to activate or suppress the enzyme.
- Competitive Inhibition: The substrate and inhibitor compete for the enzyme’s active site. With sufficient substrate concentration, maximum reaction speed can still be achieved.
- Non-Competitive Inhibition: Binds to a site away from the active site, causing a conformational change and lowering maximum reaction speed (Vmax), often irreversibly (e.g., aspirin, clopidogrel).
- Allosteric Modulation: The enzyme requires a cofactor to function (e.g., calcium in coagulation, zinc). This can either positively or negatively modulate enzyme activity (e.g., benzodiazepines with GABA).
Law of Mass Action
Law of Mass Action: The velocity of a reaction depends on the concentrations of the reactants, with equilibrium achieved at a constant relationship between reactants, defined by equilibrium constant K.
Michaelis-Menten Equation
[math] Velocity = \left( V_{max}.[S] \right) \left( K_m+[S] \right) [/math]
V = (Vmax [S]) / (Km + [S])
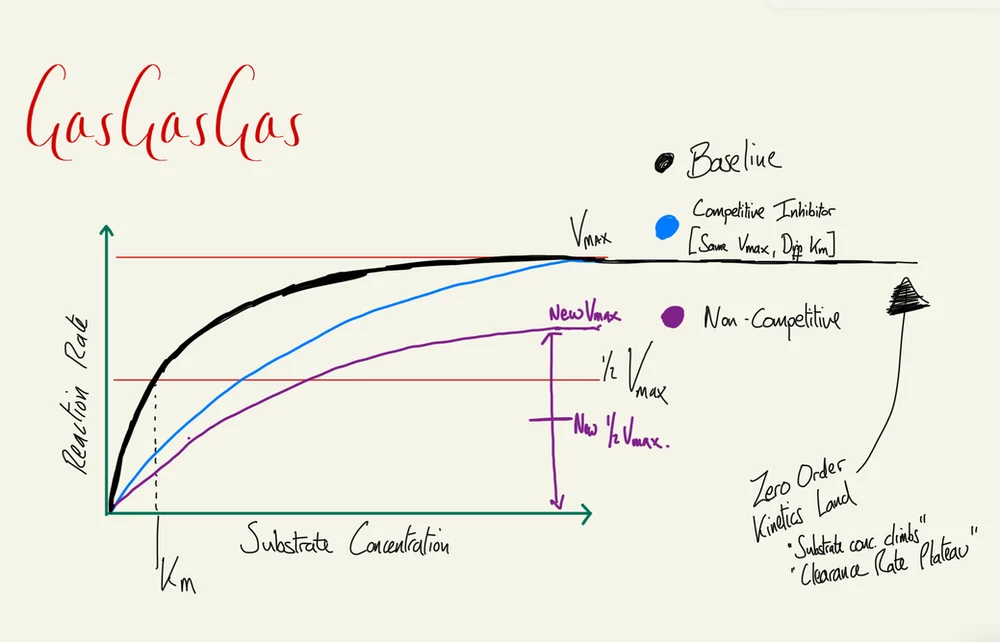
This formula applies to enzyme reactions involving weakly bonded substrates that can disassociate. Vmax is the maximum rate of the enzyme when all active sites are occupied, while Km is the substrate concentration at which the reaction rate reaches 50% of Vmax.
Core Concepts: First vs. Zero-Order Kinetics
The transition between first-order and zero-order kinetics occurs at enzyme saturation: below Km the rate scales with substrate concentration; above it the rate is fixed at Vmax regardless of further concentration increases.
- Zero-Order Kinetics: The reaction rate is constant as the enzyme is saturated; increasing substrate concentration does not increase the rate (e.g., alcohol clearance, phenytoin at toxic concentrations).
- First-Order Kinetics: A non-linear reaction rate where enzyme activity increases with substrate concentration when the enzyme system is not saturated.
Factors Influencing Enzyme Activity
- Temperature and co-factor availability
- Amount of enzyme, which can be increased or decreased with inducing or inhibiting drugs (e.g., thiopentone, phenytoin).
- Enzyme inhibitors, such as ACE inhibitors, aspirin, and neostigmine.
Graphing Enzyme Kinetics
Plotting fractional occupancy against log-concentration transforms the hyperbolic Michaelis-Menten curve into a sigmoidal curve. On a graph, the X-axis represents drug concentration on a logarithmic scale (e.g., 0-10-100-1000), and the Y-axis shows fractional occupancy on a linear scale. This provides a sigmoidal curve rather than a hyperbolic one, offering insight into enzyme kinetics across different substrate concentrations.
Summary
- At low substrate concentrations: V ∝ [S], obeying first-order kinetics.
- At high substrate concentrations: V ∝ Vmax, obeying zero-order kinetics, where enzymes become saturated, and the reaction rate cannot increase further.
References
- Hepatic Clearance — related episode on hepatic drug metabolism and extraction ratio.
Thanks for listening. Take it day by day, don't overcook yourself — keep studying.
Transcript
16 min listenRead the full transcript
Introduction and Podcast Overview
00:00-01:09
Please listen carefully. Hello, and welcome to Gas, Gas, Gas, your one-stop podcast for the FRCA primary exam. This podcast will fill your brain with information. Listen to it, think about it, and check out the show notes on the website. There you will find the core diagrams you need to be able to draw and describe for the exam.
This podcast can squeeze into your day. Listen while you’re driving to work, cooking dinner, maybe when you’re on call, or in the gym. Eventually the revision is going to end. But for now, expect facts, concepts, model answers and the odd tangent. Remember to rate and follow the show to hear much, much, much more.
Hello everyone, this is James at Gas Gas Gas. We’re here today to talk through enzyme kinetics. Going to meander through the law of mass action, what Michaelis-Menten kinetics slash the formula equation is, rumble through some definitions and make sure we’re all on the same page, and then close up with thinking about zero order and first order kinetics.
So, without further ado, we have a new subsection in today’s podcast show called “define or die,” because there’s quite a few definitions that we should go through before talking about the bulk of this. And ultimately, in the exam, if you can rumble off some definitions of the terminology you’re using, it looks pretty good.
Define or Die: Key Definitions
01:09-01:38
So, first off, enzymes. Enzymes in physiology are classically protein catalysts, and they facilitate a chemical reaction by reducing the activation energy required for that reaction. By this virtue they often increase the rate of reactions or enable a reaction that would otherwise not occur. It’s important to note that these reactions can be bidirectional.
A good example of this is carbonic anhydrase in your red blood cell swinging between carbon dioxide and water and hydrogen ions and bicarbonate, which shifts about depending on the environment the red blood cell finds itself in.
Enzyme Inhibitors
01:38-03:07
Then we’ve got enzyme inhibitors. There are several modes of action here and some other terms to think about. So a competitive inhibitor is a situation where the substrate and the inhibitor aim for the same active site on the enzyme, so they’re actively competing to bind there.
If you are in this situation, you can still achieve the maximum rate of this enzyme reaction as long as there is a sufficiently large concentration of substrate versus the inhibitor that you’ve introduced to the system - it’s competitive.
Whereas non-competitive inhibition binds to something away from the active site, inducing a conformational change in the enzyme, leading to altered binding characteristics at the active site. You can imagine here that the maximum speed of reaction that the enzyme in this system can achieve goes down as the enzyme’s a bit duff and the thing that’s meant to bind to them struggles and isn’t so good at doing it anymore.
Often in physiology these are actually found to be irreversible binders that completely dysfunction the enzyme, for example aspirin inhibiting cyclooxygenase.
Allosteric Enzymes and Law of Mass Action
03:07-04:04
There are also enzymes that require a cofactor to be bound to them in order for them to function. These are called allosteric. A number of physiological examples can be found throughout the body: phosphorylating of protein in a smooth muscle cell, the requirements for calcium in the coagulation cascade to trigger that widespread clotting cascade.
Allosteric can also be used, though, to describe how benzodiazepines positively modulate GABA activity by binding to the benzodiazepine site.
Last one is a law: the law of mass action. This describes that the velocity of a chemical reaction depends on the concentrations of the reactants. Seems pretty logical so far, and at equilibrium the concentrations of the chemicals involved bear a constant relationship to each other. This is described by the equilibrium constant, K.
The Michaelis-Menten Equation
04:04-05:28
So what is the Michaelis-Menten equation, you ask? Good question. This is an equation that describes the velocity of an enzyme-mediated reaction. It has a number of components to it.
Vmax, or velocity max, which is the maximum rate of enzyme activity when all the active sites available are active. You’ve got Km. This is the concentration of substrate needed to achieve fifty percent of the maximum velocity of the reaction.
This may sound like a familiar concept because ED50 is where you have given sufficient drug to achieve fifty percent of the maximum effect that that drug could have on a human. Km in this situation occurs at half the max velocity. Makes sense.
And then substrate concentration, S in square brackets. Square brackets means the concentration of the thing in the brackets.
Now I’m going to tell you the equation, but if you draw anything, you’re not going to remember this. So the velocity of the reaction is equal to Vmax times your substrate concentration divided by Km, that’s that half rate thingy, plus the substrate concentration. More useful if you’re doing viva practice and you can write that down.
Visualising the Michaelis-Menten Graph
05:28-06:41
This situation is better described with a plotted graph. Again, very difficult to convey with words as opposed to an image. It’s better to have a look at this. But generally speaking, you’re looking at a graph with substrate concentration plotted along the x-axis and rate of reaction like zero to a hundred or zero to one on the y-axis.
It follows a neat smooth curve with a steep initial aspect and flattening off towards the end. This is because as you add more and more substrate concentration, there’s very few enzymes that are actually free to act, because most of them are tied up at this point in time. So you end up with diminishing returns with greater substrate concentrations.
And this is why Km, that half the velocity of the reaction rate point, might actually occur at a fairly low substrate concentration, especially if the enzyme has a high affinity for the substrate. You could imagine at this point if you had an enzyme that had a low affinity for the substrate, i.e., perhaps wasn’t terribly efficient, you would need relatively higher substrate concentrations in order to achieve full effect. That curve may look a bit different.
Effect of Inhibitors on the Graph
06:41-07:36
Your brain says, “hang on, but you’ve talked about competitive and non-competitive enzyme inhibitors at the start of this,” and you’re right. If we return to imagining a graph that has a fairly steep initial aspect of the curve that then plateaus off, and we were to introduce a competitive inhibitor to the system, then that curve would still reach maximum enzyme reaction velocity eventually, but it’s going to be a bit flatter. It’s not going to be as steep at the start because there is competition between inhibitor and substrate for the enzyme.
The next step is if you add a non-competitive inhibitor to this situation, your maximum reaction velocity will go down because it’s just bound to some enzymes and duffed them up and they don’t work anymore, so they can’t get involved, and therefore the reaction rate slows, or at least the maximum reaction rate diminishes.
Zero Order and First Order Kinetics
07:36-09:14
The next logical step in thinking about this graph is what occurs when you just keep adding substrate and piling it in buckets, buckets of the stuff. Well, eventually all your enzymes are working away, going hell for leather, and they can’t work any faster, so you accumulate substrate.
This situation is where zero order kinetics occurs, i.e., the rate of enzyme activity or the rate of clearance of the thing that needs to be cleared or produced, becomes fixed because there’s abundant substrate and the rate limiting factor becomes the enzyme.
The contrary to this is in a situation where you have abundant enzyme and less substrate availability, you can imagine that those enzymes are going to rapidly mop up and hoover up, and then the enzymes are going to end up competing for the substrate. This leaves us in a situation where, as there is fewer and fewer substrate available, it’s less and less likely to bump into enzyme, the rate of reaction diminishes in a nonlinear manner. This is first order kinetics, that is, it’s nonlinear.
Just to revise that again, guys, remember, so zero order kinetics is a saturated clearance situation already - saturated enzyme binding site situation, whereas first order kinetics is where the situation is not saturated and the enzymes are actually gagging for substrate, but there’s not very much knocking around.
Clinical Examples
09:14-10:25
I’ve touched on in previous podcasts that this concept of first and zero order applies to clearance in systems as well. Good examples of drugs that exhibit zero order kinetics: phenytoin rapidly saturates the enzyme clearance system it is associated with, and alcohol - that is why alcohol is measured in units. Units are the amount of alcohol cleared by a standard human in an hour. That’s a fixed rate of clearance now, isn’t it?
I’m sure no one recalls the situation as being terribly, terribly hungover and then suddenly feeling quite a bit better just at the tail end of everything. And that’s because that process of clearance has rapidly tidied up, the last remnants having been at a fixed rate of declination for a while.
A good example of the opposite, i.e. a first order system that is very challenging to saturate, is your remifentanil and your plasma esterase system. Because the plasma esterase system is abundant and therefore it’s quite hard to saturate it, and that’s why there is no such thing as the context sensitive half-time almost in remifentanil. Obviously, it has one, but it’s so short relative to if you were to just pour fentanyl into a human being over a long period of time.
Check out the Volume of Distribution podcast to learn more about fentanyl.
Factors Influencing Enzyme Activity
10:25-11:52
Now, if you’re in the exam, they might also tip a little bit closer towards just biochemistry on this one and ask you, “Doctor, what factors influence enzyme activity?” This is quite simple. You could break it down into physical and physiological.
So physical things are what is the temperature of the enzyme system? If you’re thirty degrees your enzymes are working slower. If you’re forty-two degrees your enzymes are working slower. What cofactor availability is there for your enzyme? Is there enough zinc floating around? And whether or not there’s any presence of inhibitors, either physiological or iatrogenically introduced, for example, ACE inhibitors, aspirin, neostigmine.
Physiologically, it’s important to think about enzyme-inducing or inhibiting drugs, those which actually alter hepatic enzyme synthesis, i.e. do they jazz it up or do they calm it down?
Your classic jazz it up drugs: barbiturates and your thiopentone, carbamazepine, alcohol consumption over time, phenytoin, isoflurane and halothane have been implicated. Smoking induces certain enzymes, which then mean you clear sevoflurane better, if I remember correctly.
Whereas the inhibitors you might choose to talk about, I would mention amiodarone, erythromycin, omeprazole, and grapefruit.
Drawing Graphs in Exams
11:52-12:49
The last thing to say about all of this, which is relevant to the viva, is when you are drawing out a graph and plotting those axes, do it in an organised manner. So for that graph I mentioned earlier, the x-axis is the concentration of drug or substrate. This is logarithmic for the Michaelis-Menten enzyme kinetic graph, i.e., the x-axis goes zero, ten, one hundred, one thousand, ten thousand, beyond.
The y-axis is the fractional occupancy rate. This is linear. It’s either zero percent occupied, one hundred percent occupied, or somewhere in between. The reason this is done is because you could imagine otherwise you would lose quite a lot of information at the extremes of enzyme activity because of those diminishing returns that you might experience on either end. This gives it a nice sigmoidal curve instead of that hyperbolic curve.
Summary
12:49-13:21
So to summarise, probably the most important thing to grasp in enzyme kinetics is that in a low substrate concentration environment, most systems obey first order kinetics, and as the substrate concentration increases, so does the rate of the reaction, whereas at high substrate concentrations, your velocity becomes fairly equivalent to Vmax and it obeys the zero order kinetic situation because everything’s saturated, it can’t go any quicker.
Conclusion
13:21-13:45
If you found it useful or awful, please like and subscribe and rate the show. Definitely check out the show notes for those diagrams and the detail of this content. It is a bucket of content to get to grips with. Keep working at it and you will get better, faster and stronger. It is vital to keep your interest alive for the science that we’re covering and not overcook yourself. You will be amazed by what you know come exam day. Don’t freak out, keep studying.
Enjoyed this? Review on Apple Podcasts Rate on Spotify
Support the show Help keep the lights on SBA question bank @ Teach Me Anaesthetics
