Mivacurium For Anaesthetists
1 February 2026
Introduction
Mivacurium is a non-depolarising neuromuscular blocking agent of the bis-benzylisoquinolinium class, with a unique metabolism by plasma cholinesterase considering it is a non-depolarising muscle relaxant. Developed to provide more stable cardiovascular side effect profiles than its prototypes with predictable, context-insensitive offset, mivacurium offers a shorter duration of action than atracurium or rocuronium whilst avoiding the side effects of repeated suxamethonium dosing.
Mivacurium represents an important comparison point: understanding why its duration remains relatively constant regardless of dose (first-order kinetics with non-saturated enzymatic clearance) demonstrates key pharmacokinetic principles. Its metabolism pathway also creates a direct link to plasma cholinesterase deficiency – candidates who understand suxamethonium apnoea can apply the same reasoning to predict mivacurium’s behaviour in affected patients.
This episode explores mivacurium’s physicochemical properties, its three-isomer composition, and the practical pharmacokinetics that made it an attractive option for short procedures, once upon a time.
Mivacurium Pharmacology
The following sections cover physicochemical properties, pharmacodynamics, and pharmacokinetics — structured to match the FRCA Primary pharmacology viva format.
Mivacurium Physico-Chemical properties
| Name | Mivacurium |
| Class | Bis-Benzylisoquinolinium diester – Bis means ‘twice or two’ |
| Chemical Make Up | C58H80N2O14+2 |
| History | A goal in its development was to have a cardiovascularly stable neuromuscular blocker (NMB) that was context insensitive (any dose leads to near same block duration) without the side effects of high dose or multi dose (suxamethonium for example with its type II block.) Benzylisoquinilinium paralytics were initially discovered in 1961 – RT Brittain, HOJ Collier, PF D’Arcy with a curare like molecule called γ-oxalolaudonium bromide – progress was made from these earlier agents towards mivacurium (excesses of histamine release were an issue, and this first molecule provided very brief paralysis in cats….) |
| Isomer Status | A mix of three stereoisomers – -trans, cis-trans and cis-cis cis-trans and trans-trans (37% and 57% of the ampoule) are mostly equipotent. cis-cis – 6% of the ampoule markedly reduced potency – 1/10th of its pals. |
| Colour/Appearance | A clear, pale yellow aqueous solution 5 and 10 ml amps, 2.14mg/ml of mivacurium hydrochloride pH 4.5 |
| Molecular weight | 1029.3 g/mol!! Quite ‘heavy; compared to some other NMBs, its molar potency is therefore different (roc 529, suxamethonium 361, trac 929) |
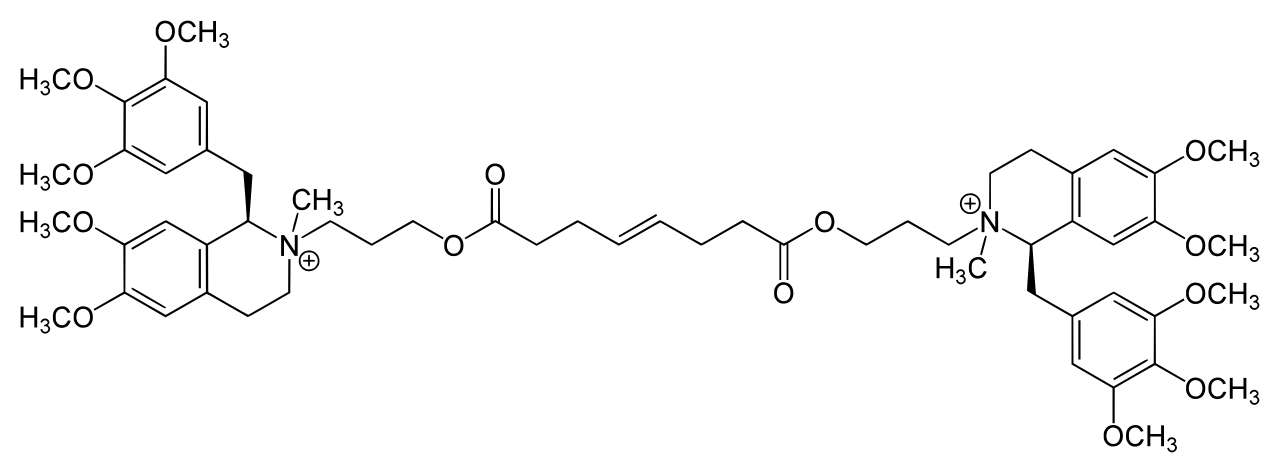
Mivacurium molecule, note the C=C double bond but also the nature of the ring systems and its ability to ‘up or down in and of itself’ around the c1 point adjacent to the nitrogen molecules.
Also spot the esters, where the carbon back bone has a double bond to an oxygen. These are along from either side of the central carbon double bond.
Mivacurium Pharmacodynamics & Side Effects
| Mechanism of Action | Competitive non-depolarising neuromuscular blockade (Check out rocuronium for more details) |
| Chief Effect / Actions | Antagonism of acetylcholine at the nicotinic (N2) receptor situated on the post synaptic membrane of the skeletal neuromuscular junction |
| Dose | Intubating – 0.2mg / kg – IV – Onset :~2 mins Maintenance – 0.1mg/kg/15 mins ED95 for paralysing dose in one study – 0.08mg/kg – 3.8 min onset time Duration is longer than suxamethonium, 15-20 mins of effective blockade. But shorter than atracurium and rocuronium. Paediatric patients average duration for standard (0.2mg/kg) dose = 6-7 mins of useful clinically effective block Onset time is similar to that of Atracurium at clinical doses used for both (i.e. you could use a 0.1mg/kg dose of mivacurium, but most folks use a bit more so there is less waiting) |
| Cardio-Vascular Side Effects | Headline : Histamine Release from Mast cells > Flushing and Hypotension in around 15% of patients Chronotropy : +ve (‘7%’) |
| Respiratory Side Effects | Rate – Obliterated Depth – Obliterated Parenchymal effects: BronchoSpasm Trigger (Histamine release) – So don’t mix it with asthmatic or airway hypersensitive patients. and probably those with Mast Cell Activation Syndrome for good measure. |
| Central Nervous System Side Effects | |
| Metabolic/MSK: Side Effects | Transient Cutaneous flushing (Histamine) Effect extended by volatile anaesthetic agents |
| Toxicity | If a patient has sux apnoea due to cholinesterase deficiency – they will also have ‘miv apnoea’ |
Mivacurium Pharmacokinetics — Context-Insensitive Offset
| Absorption | n/a |
| Distribution | Volume of distribution 0.2 L/kg (big, polar molecule, doesn’t exactly float off into adipose tissue) Volume of distribution varies by molecule (likely aligning with the rate of clearance/ half-life) Trans-Trans 147ml/kg Cis-Trans 276, Cis-Cis 335 |
| Metabolism Remember: Phase I : [oxidation, reduction, hydrolysis] (more cytochrome action here) (more O2 needed) occurs in inner aspect of liver acinus… Phase II: [conjugation, glucoronidation, acetylation, sulphylation] (less O2 needed) occurs in outer aspect of liver acinus… | Enzymatic hydrolysis by PLASMA cholinesterase (butyrylcholinesterase) Comparative rate of catalysis 88% the rate of suxamethonium clearance. Yielding a quarternary alcohol and monoester inactive metabolites. Behaves in a first order manner, given the similar return of muscle power with varying dose durations, implies non saturated enzymatic clearance (check out the michaelis menten enzyme kinetics episode) Is reversible with [neostigmine](/episode/anticholinesterases/) + an anticholinergic if the surgeon plucks the offending lesion off in a snap. Paediatric cohort appears to metabolise it quicker Endstage hepatic failure is associated with ~1 hour of block, cirrhotics, 45 mins. Those with nephrotic flavoured renal failure will be whizzing out anaesthetically useful proteins and have a degree of block prolongation also. |
| Elimination | Overall Half-life of 1.8-2 minutes Half-life Per Isomer cis-trans 2.1 minutes trans-trans 2.3 minutes cis-cis 55 minutes (remember its only 6% this) Clearance to quote overall : 4.2 L/kg/min (when given as bolus) Clearance Per Isomer (as an infusion) cis-trans 5.5 L/kg/h trans-trans 3.2 L/kg/h cis-cis 0.25 L/kg/h |
Mivacurium in Practical Anaesthesia
Mivacurium’s context-insensitive offset, dose-response plateau, and sensitivity to plasma cholinesterase status are the three practical considerations that dominate its clinical use.
Mivacurium dose:Response
ED95 = 0.081mg/kg
0.1mg/kg =95.7% of patients blocked – recovery taking 24.5 ±1.6 mins.
0.2mg/kg = 30 min of block
0.3mg/kg = 36 mins of block
So significantly diminishing returns despite 2x and 3x doses! but as you would expect a faster onset of action is noted.
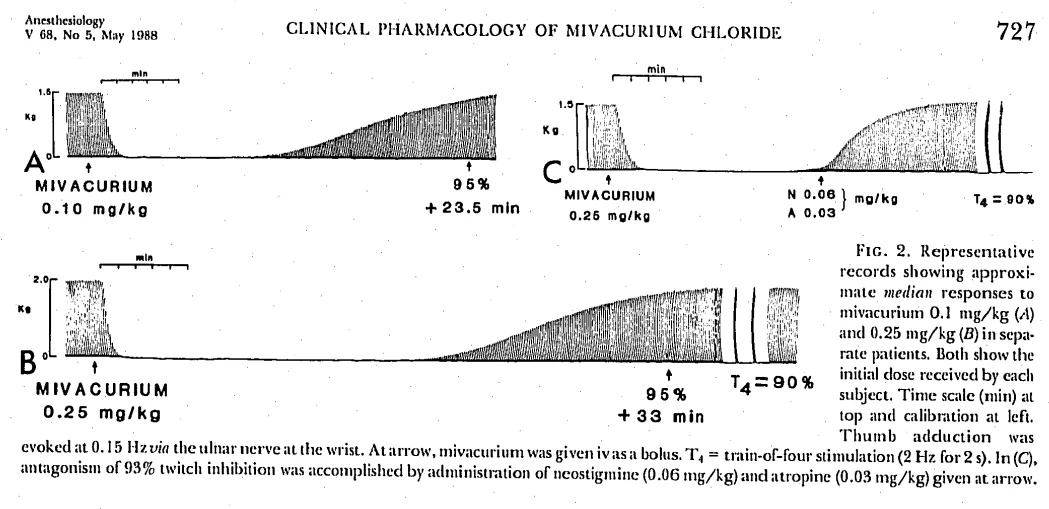
Mivacurium block recovery times with varying dosages (ref below)
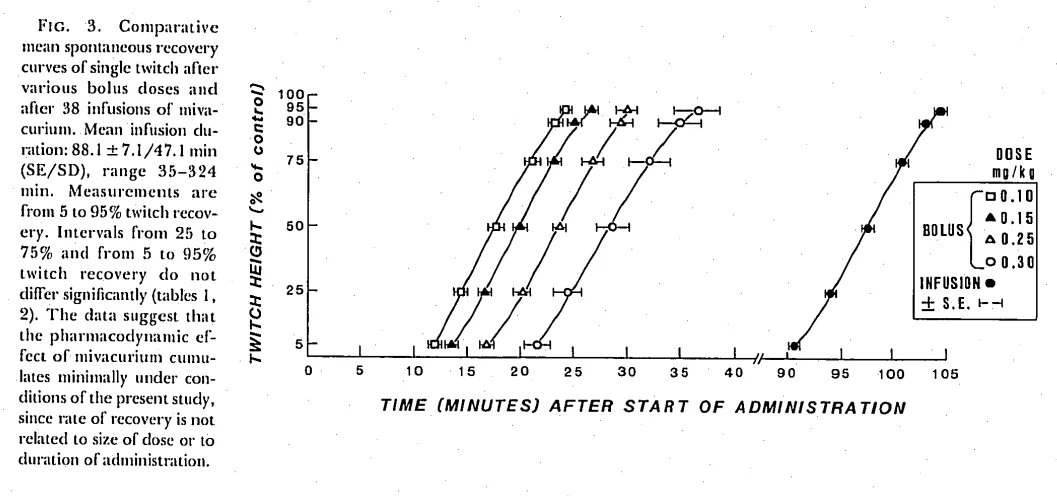
This demonstrates the very similar curve of recovery from a mivacurium infusion of differing durations (ref below)
Isomers revisited
Cis-trans isomers are a sub-type of stereoisomer called geometric isomers.
They have the same molecular formula and general layout with a differing spatial arrangement that pivots around a fixed (no rotation can occur) point in that molecular layout, more often than not this pivot is a c=c carbon double bond.
About this point the molecule can have one ‘face up’ and one ‘face down’ side, be both face up, or both face down.
Different morphology confers different properties, be that receptor affinity or lipophillicity (more or less polar)
Remember cis = the r group or equivalent is on the same side of the double bond
trans = the r group is on the opposing side of the double bond
Mivacurium Apnoea
Miv Apnoea is a possibility, given its relative dependence on plasma cholinesterase, if you’ve a patient who has a paucity of this enzyme – mivacuriums effects will be prolonged….
It is suggested that it won’t last quite as long as suxamethonium in this situation, given that 1mg/kg of suxamethonium is 4-5x the ED 95 and thus there will be quite a bit more to get rid of before muscle power return. Further to this, it may well be that there are other routes of mivacurium metabolism (liver) as noted in cat and dog models….
Reports in the early nineties found some episodes of mivacurium apnoea lasted for 3-4 hours. If you’ve a patient homozygous for atypical plasma cholinesterase, pick something else (bar suxamethonium!)
Suxamethonium Apnoea – AKA PseudoCholine-Esterase Deficiency
Plasma Cholinesterase activity is influenced by genetics and / or acquired factors. nb pseudocholinesterase is a old fashioned name….
When there is significant enzyme dysfunction, the state shifts effectively into a zero order process as the system is saturated by masses of suxamethonium, and scant or desperately feckless plasma cholinesterase enzyme. Leading to prolonged effect of drug!
Enzyme coded for on Chromosome 3
| Allele / Name of Enzyme | Incidence | Offset |
| Normal / Normal (Eu) | 96% of population normal. | Normal |
| Normal / Abnormal | Almost 4% of the population | 10 mins |
| Atypical (dibucaine resistant) | 0.03% (3:10000) | Hours |
| Silent | 0.001% (1:100 k) | Hours |
| Fluoride Resistant | 0.0003% (3:1 mil) | Hours |
Identification: if you TOF everyone post suxamethonium then you might find a few heterozygous atypicals…
Treatment = Keep the patient sedated and avoid the Trifecta of Badness.
- Don’t wheel them around the hospital for a CT scan thinking something intra-cranial happened…..
- With no sedation aboard
- Whilst also forgetting to check the TOF.
You could transfuse FFP, providing pseudocholinesterase and making it all better, but most of the time a wait occurs – I don’t know what the plasma HL of pseudocholinesterase is, but seeing as once its gone its gone, the suxamethonium probably couldn’t re-paralyse the patient….. bold to send them home.
They will eventually pee it out….
Send them for testing + refer them to clinical genetics.
Just one more thing!
I’ve said that a geometric isomer is cis or trans, but have also told you that there is a cis-cis variant of mivacurium. If a molecule has more than one fixed point around which a gross layout can change – then you find yourself using multiples of cis or trans to describe it.
Summary
Mivacurium is a bis-benzylisoquinolinium non-depolarising neuromuscular blocker metabolised by plasma cholinesterase at 88% the rate of suxamethonium. Its three stereoisomers (trans-trans 57%, cis-trans 37%, cis-cis 6%) have markedly different half-lives – the cis-cis isomer persists 25 times longer than its counterparts but contributes minimally to clinical effect due to its reduced potency.
The defining feature of mivacurium is context-insensitive offset: whether you give 0.1 mg/kg or 0.3 mg/kg, recovery time increases only modestly (15 to 36 minutes), reflecting likely non-saturated first-order elimination. This contrasts with agents dependent on redistribution, spontaneous breakdown (atracurium) or hepatic metabolism (rocuronium).
Clinically, remember three things: histamine release causes hypotension, flushing and bronchospasm (avoid in asthmatics), plasma cholinesterase deficiency prolongs block dramatically (consider alternatives in known or suspected cases), and neostigmine can reverse residual block if needed. Sugammadex does not reverse mivacurium — see sugammadex for the aminosteroid reversal contrast. The ED95 is approximately 0.08 mg/kg, with standard intubating doses of 0.2 mg/kg providing 15-20 minutes of effective paralysis.
References
- Savarese, John J. M.D.; Ali, Hassan H. M.D.†; Basta, Salvatore J. M.D.‡; Embree, Patricia B. C.R.N.A.§; Scott, Ralph P. F. M.D.‖; Sunder, N. M.D.‡‡; Weakly, J. Neal Ph.D.*; Wastilla, William B. Ph.D.††; El-Sayad, Hassan A. Ph.D.‡‡. The Clinical Neuromuscular Pharmacology of Mivacurium Chloride (BW B1090U). A Short-acting Nondepolarizing Ester Neuromuscular Blocking Drug. The Journal of the American Society of Anesthesiologists 68(5):p 723-732, May 1, 1988. | DOI: 10.1097/00000542-198805000-00010
- Frampton, J.E., McTavish, D. Mivacurium. Drugs 45, 1066–1089 (1993). https://doi.org/10.2165/00003495-199345060-00009
- H.H. ALI, J.J. SAVARESE, P.B. EMBREE, S.J. BASTA, R.G. STOUT, L.H. BOTTROS, J.N. WEAKLY, CLINICAL PHARMACOLOGY OF MIVACURIUM CHLORIDE (BW B1090U) INFUSION: COMPARISON WITH VECURONIUM AND ATRACURIUM, British Journal of Anaesthesia, Volume 61, Issue 5, 1988, Pages 541-546,
- Brittain RT, Collier HO, D’Arcy PF. The neuromuscular blocking action of gamma-oxalolaudonium bromide. Br J Pharmacol Chemother. 1961 Aug;17(1):116-23. doi: 10.1111/j.1476-5381.1961.tb01111.x. PMID: 19108153; PMCID: PMC1482061.
- Meakin GH. Neuromuscular blocking drugs in infants and children. BJA Education 2007. https://www.bjaed.org/article/S1743-1816(17)30443-2/fulltext
Thanks for listening. Take it day by day, don't overcook yourself — keep studying.
Transcript
25 min listenRead the full transcript
Mivacurium – Gas Gas Gas Podcast Transcript
Introduction
00:00-00:58
Hello, Team Anaesthesia. Welcome to Gas, Gas, Gas – the best anaesthetic science podcast for the FRCA Primary exam. Our goal is to fill your brain with all this highly useful information. Now, you might be in the gym right now, commuting, or ironing your scrubs, and there’s no judgment here. Gas, Gas, Gas will prime your brain for the monsoon of knowledge you need to imbibe. But regardless, the revision is eventually going to end. But for now, expect facts, concepts, model answers, and the odd tangent. Now remember to check out the website, that’s gasgasgas.uk. There are show notes there with all the detail, plus links to foundational reference papers and anything else useful I find for you guys. Anyway, buckle up, get ready for your mind to be bent into a new shape, and let’s get on with the show.
Episode Overview and Future Content
00:58-01:56
Welcome to Gas Gas Gas. We’re talking mivacurium today. You may have seen it be used once or twice. These interesting properties also mean that if it were being used in clinical practice, it carries some benefits and some drawbacks. So before I get into the bulk of this episode, I just want to give you a heads up for where things are going to be going in the next few months, Gas, Gas, Gas wise. I want to handle the rest of the neuromuscular blocking drugs because we’ve done suxamethonium, rocuronium and atracurium, which are the common ones. But also, someone in your department might still like using vecuronium. There is cisatracurium, which again, you might find in the cupboard. It might get used once or twice if you’re in a teaching hospital where they actually decide to buy it. And then we’re going to do pancuronium just because that’s interesting. Then we will handle paracetamol, non-steroidals, and coxibs before scoping out a bunch of episodes where we handle all the things that can make your heart rate better, your heart more squeezy, and your blood pressure less saggy.
Key Headlines and Classification
01:56-03:27
Anyway, mivacurium. What are the headlines that you should know and we’re going to cover in this episode? It behaves in a first-order kinetic manner. Why is this? Because it is broken down by plasma cholinesterases like suxamethonium. And there are three isomers of mivacurium. Again, why it becomes tempting for a conversation about stereoisomers, etc. in an exam setting.
Benzylisoquinolinium diester. What does the ‘bis’ mean? The ‘bis’ is two because it’s sort of a two-ended molecule, a bit like suxamethonium. A diester because it’s got two esters, although technically it has four. And benzylisoquinolinium is really what you should try and remember to cough out in the exam if you asked to classify neuromuscular blocking drugs, because you would say depolarising and non-depolarising. Depolarising: suxamethonium. Non-depolarising: well I like to break this down into aminosteroid non-depolarising muscle relaxants and benzylisoquinolinium non-depolarising muscle relaxants. And then you’d say aminosteroids: vecuronium, rocuronium, pancuronium. Benzylisoquinoliniums: atracurium, cisatracurium, mivacurium. You do that, you’re well on your way to making good progress in that question.
History of Mivacurium
03:27-04:26
What about a little bit of the history of mivacurium? Hard to find explicitly on the interwebs, but benzylisoquinoliniums were initially discovered in 1961, where Breton, Collier, and Darcy were mucking around with a curare-like molecule called gamma-oxalolaudonium. Now, laudanosine – think laudanosine – this is a metabolic byproduct of atracurium and mivacurium, coming to that later, but they noted that giving this to cats caused very transient paralysis, but also excesses of histamine release. So the goal was to try and develop a benzylisoquinolinium that caused less histamine release and lasted a bit longer, as you might expect. Although I imagine the suxamethonium runs with gamma-oxalolaudonium bromide would be very brief, perhaps a bit safer, who knows, I don’t know if humans ever got it. So with mivacurium they actually did a great job and created a context-insensitive, infusible without too many side effects drug in mivacurium. Remember once upon a time when you needed to keep someone paralysed, folks were trying to do suxamethonium infusions and all sorts, several side effects to that.
Isomer Status
04:26-04:55
What is mivacurium’s isomer status? There is a trans-trans, a cis-trans, and a cis-cis. If you have an ampoule of mivacurium in front of you, only 6% of that ampoule is cis-cis mivacurium. So it doesn’t work very well, and there’s not very much in the ampoule, which is kind of what you’d hope for. Whereas the other two, your cis-trans and your trans-trans, 37% and 57% of the ampoule respectively, and they are pretty much equipotent.
Physical Properties
04:55-06:10
Colour and appearance. So mivacurium is presented as a clear or sometimes pale yellow aqueous solution in 5 or 10 ml ampoules containing 2 mg per ml of mivacurium chloride. And its pH is 4.5, so a bit acidic and stingy.
Its molecular weight. Now, I’m sure this is the brief moment of the episode where you fall asleep, but actually, prepare to be stunned, mivacurium is really quite heavy. It is 1029.3 g per mole, so a chunking molecule. Remember, rocuronium only 529 and suxamethonium 361, so it’s twice as hefty as rocuronium. Atracurium is not far behind mivacurium at 929 grams per mole, and this in your mind fits because mivacurium and atracurium are benzylisoquinoliniums, and you need to practise saying that so it just rolls off your tongue.
Understanding Geometric Isomers
06:10-08:19
Now we’re going to get into isomers more heavily after the main pharmacological bulk, but remember that a geometric isomer is where you have arms of a molecule or arms of a molecular structure that can alternate around a fixed bond. I.e. there’s a fixed point, classically a carbon double bond, that allows an arrangement to be one way or the other on the opposing aspects of that bond.
Now, classically, this is taught as the old-school cis-trans nomenclature. You either have both of your R groups, or two identical R groups on opposite sides of the bond, or you have them on the same side of the bond. And this classically can be thought of with our trans fats. So we’ve all been told that trans fats are bad, and trans fats should be avoided. And trans fats, essentially, with that double bond all lined up nicely, they are very efficient to pack, and therefore they pack in very tightly, which makes them solid at room temperature, which therefore makes them much more useful for baking and giving you that crispy pastry texture. Cis fats, on the other hand, are generally not solid at room temperature, and are much less well-packed. And also, our biological machinery can much more readily metabolise cis fats. So cis fats would be your unsaturated fats, your vegetable oils. Trans fats would be your saturated fatty acids, and these are your animal fats, butter, processed foods. So, if you think your trans fats are bad, that may therefore help you remember trans-trans mivacurium is bad, sort of, it’s feckless, it’s not really doing much.
Clinical Pharmacology
08:19-10:20
So, what about the clinical side of things? What’s the ED95 dose? Classically, the dose that you should be giving is two times that ED95, and that is going to be in the region of 0.2 mg per kg, or put in another way, you’re going to be giving around 15 mg, and it’s going to produce a neuromuscular block that’ll last around 15 to 20 minutes at that 0.2 mg per kg dose. The onset is going to be between 2 to 3 minutes.
The duration is very dependent on that dose, and you’ve got to think about how this is being cleared. If you give them 0.15 mg per kg, you’re going to get block for about 12 to 18 minutes. Give them 0.2 mg per kg, you’re in the region of 15 to 20 minutes. Give them 0.25 mg per kg, you’re going to be sitting around between 20 to 25 minutes before things have recovered. And what do we mean by duration? What we’re always talking about is the time to 25% recovery of your train-of-four count. And the train-of-four count is really useful for monitoring the degree of paralysis.
A standard intubating dose is around about twice the ED95, so classically this is 0.2 mg per kg or approximately 15 mg. Again, depending on patient weight, it might be more or less. And the redistribution is as with other non-depolarising neuromuscular blockers, so it redistributes away from the neuromuscular junction.
Mechanism of Action
10:20-11:17
The mechanism of action of mivacurium. Remember, it is a competitive antagonist at the nicotinic acetylcholine receptor on the post-junctional membrane of the neuromuscular junction. It will compete with acetylcholine at that post-junctional receptor. And this therefore inhibits depolarisation, stopping muscle contraction. Now, remember you need around 75 to 80% of those receptors blocked before twitch height starts diminishing and clinical effects become apparent. Non-depolarising drugs, so this is mivacurium as well, cause what we call a fade where you stimulate the muscle at an increasing frequency, you get less and less of a twitch. And this is very different to suxamethonium where you stimulate the muscle very, very quickly and you still get the same twitch height. And that’s important.
Metabolism and Clearance
11:17-13:14
Its metabolism. This is one of the key things to remember for mivacurium, the reason it’s clinically relevant. Mivacurium is hydrolysed by plasma cholinesterases to inactive metabolites. And that’s monoester mivacurium and then also to the quaternary alcohol and the monoester quaternary amine. However, there is a small amount, probably in the region of around 10%, that spontaneously degrades to laudanosine. We’ve mentioned laudanosine before. At very high doses, and you’re really not going to get this with anaesthetic practice, it can cause CNS excitation and possibly even seizures.
Now, the hepatorenal clearance of mivacurium is minimal. And this is really different to a lot of other drugs, but the same as suxamethonium in that actually, providing your enzymes are working, even if your kidneys and your liver are both really quite poorly, you can still give mivacurium and it’s still going to be cleared. And this is clinically useful.
The elimination half-life varies. So for your trans-trans and cis-trans isomers, which are the most active ones, you’re looking at around 2 minutes. But the cis-cis, your feckless variety, is going to be much longer, around 50 to 70 minutes. But again, remember, there’s not very much of it there anyway. And this therefore is reflected in your time to 95% recovery of twitch height. In the population in general, it’s going to be about 25 to 30 minutes. So by the time they’re in recovery and you’re hassling the ODPs, it’s about 25 to 30 minutes until they’re back to 95% normal again. Duration of infusion has little effect on recovery time, and this again is very different to other muscle relaxants. It is a context-insensitive drug, which is actually probably its single saving grace. All the other reasons why it’s not used mean it doesn’t get used very much at all.
Cardiovascular and Other Effects
13:14-15:17
Its cardiovascular effects. It has minimal cardiovascular effects at normal doses. However, it can cause histamine release. At doses above 0.2 mg per kg, you start to see significant histamine release, which can cause flushing, hypotension, bronchospasm. And you don’t want your asthmatic being a little bit wheezy, then give them some mivacurium and then have them very wheezy.
It has no ganglion-blocking effects, unlike some older neuromuscular blockers. So your pancuronium actually has a muscarinic effect. It blocks muscarinic receptors, which therefore means folks get tachycardic when you give them pancuronium. That’s not really a huge problem for most people, but it can be.
Other effects. CNS penetration – it doesn’t cross the blood-brain barrier. It’s a large, charged molecule. It can’t get into the brain. Likewise, it doesn’t cross the placenta in clinically significant amounts. Remember placenta is also not very fond of charged, heavy molecules. So it’s safe to use in obstetrics theoretically, although again, not commonly used. And there’s no analgesic properties. This is a muscle relaxant. It doesn’t take pain away.
Train of Four Question
15:17-17:47
Mivacurium Apnoea
17:47-20:47
Mivacurium apnoea is a possibility because mivacurium is cleared by plasma cholinesterases, and folks in the population have varying qualities of plasma cholinesterases. 96% of people are knocking around with normal, happy-go-lucky plasma cholinesterases that can spot a suxamethonium molecule from a mile away and, you know, sufficiently duff it up that its mischief stops quite quickly. However, 4% of the population have one normal gene to code for plasma cholinesterases and one abnormal gene, almost 4% of the population. It’s like almost 1 in 20 of your patients. And you will see a slightly prolonged effect of drug. Quoting it for suxamethonium, suxamethonium lasts between 5 to 7 minutes or so. It would take 10 minutes.
And then there’s a subset of the population where there’s very few of them, thankfully, who have either atypical, silent or fluoride-resistant coded plasma cholinesterases. And out of those, it’s 3 in 10,000 folks are atypical. One in a hundred thousand are silent and three in a million are fluoride-resistant. It is inheritable. That’s why if you have a patient with suxamethonium apnoea you can refer them to clinical genetics.
If you imagine our situation where you’ve gone from happy-go-lucky, they’ve had their coffee enzymes that are clearing suxamethonium like no tomorrow to some quite feckless, you know, sleep deprived. Maybe they’ve got a newborn at home, these enzymes, and they’ve not really had much sleep and they don’t process drug as quickly. Those are effectively either slow enzymes or now and subsequently leads to a saturated clearance system. So you can end up well away into that first-order Michaelis-Menten zone whereby it takes a really long time to clear drug.
Now everyone can recall suxamethonium apnoea quite happily can’t they? But mivacurium some people forget and you might find yourself in the trifecta of badness. What is this trifecta of badness? Well it’s when you wheel a patient around the hospital for a CT scan thinking they have had an intraoperative, intracranial event, you’ve not put sedation on them and you’ve not checked their train-of-four. And then you subsequently realise they have suxamethonium apnoea and you’ve given them mivacurium. Apparently this may have happened in the region I work in all that time ago when mivacurium was more readily used. If someone ain’t waking up and you’ve not already checked their train-of-four, check their train-of-four. Don’t be a lemon.
Managing Mivacurium Apnoea
20:17-21:17
What do you do? If you realise they’re still paralysed, talk to them in a very kind and gentle manner. Tell them you’re going to give them some medicine to make them feel sleepy and that you’re going to take excellent care of them and that they will be okay. And then you have some choices. You could give them FFP and this would provide them with plasma cholinesterases from somebody else that odds are will work and you’ll then clear that pesky drug. Or you can park them on intensive care and wait.
Now transfusing FFP sounds like a quick and excellent solution and a nice interventionist approach and we like to do stuff. But then we expose the patient to blood products, perhaps unnecessarily. The challenge becomes what if they have really, really, really lazy enzymes and they’re then subsequently stuck on ITU for days and days and days, although unlikely. You then need to consider the implications of being on a ventilator for several days, the increased risk of venous thromboembolism, the increased risk of ventilator-associated pneumonia, the loss of muscle power, etc., etc., that comes with that. Don’t forget to send them to genetics afterwards.
Isomers Revisited
21:17-23:18
Let’s just quickly revisit isomers. Remember, an isomer is something with the same formula, but a different set of atomic connections with differing physical properties. There are a multitude of types of isomers. Structural and stereo with subtypes beneath them.
So structural isomers are where your functional group or an R group is in a different position on that carbon backbone in organic chemistry. You know, functional isomers where they’re actually completely different molecules but with the same molecular makeup. Tautomers, which aren’t technically isomers, whereby an environmental change alters the shape of that from an open ring to a closed ring type structure.
And then you have stereoisomers. Remember these are same formula and same chemical structure but different three-dimensional arrangement. And we can break this down into our geometric isomers and our enantiomers. Mivacurium falls into the geometric isomer realm. And this is our cis-cis, trans-trans, cis-trans nomenclature. Our enantiomers are non-superimposable mirror image molecules. This is our levo-dextro and our sinister-rotary. Bupivacaine is the one to really describe that.
However, there’s also another stereoisomer friend, and that is our diastereoisomers. These are drugs with more than one chiral centre, and therefore you can have a bunch of different configurations. And atracurium, our friend atracurium, has a mere 10 isomers because it has four chiral centres. For more joys about isomers and a thorough conversation about it, check out episode 39 called Isomers.
Now, much like you can have diastereoisomers with more than one chiral centre, you can also have more than one fixed point around which a molecule can rotate. Mivacurium is such this. It actually has four fixed points that can vary because surely a cis-cis variant doesn’t seem to make sense does it? You either have cis- or trans-mivacurium. How can you have cis-cis-mivacurium or trans-trans-mivacurium? And that’s because if you imagine a molecule with four points that are fixed but confirmations that can change around all four points you have to have some way of describing it.
Summary and Exam Tips
23:18-24:07
Mivacurium. We’ve had quite an extensive chat. We don’t use it very much anymore. The important things to remember for the exam is that you can use it as an example of cis and trans geometric isomerism. It relies on plasma cholinesterases so you can get mivacurium apnoea. It is relatively context insensitive because of this enzyme system that degrades it and it causes histamine release. So in an exam where you’ve had a patient who’s got really you know ticklish asthma, brittle asthmatic if you want to use that label, you wouldn’t pick mivacurium and actually rocuronium is probably the best one to choose.
And my last top tip is do not confuse non-specific plasma esterases with plasma cholinesterases because they are different. Plasma esterases you will find get busy with remifentanil, remimazolam and esmolol.
So I hope you enjoyed that episode. If this is the first episode of Gas, Gas, Gas you’ve ever listened to, congratulations. There’s so many more you can listen to in that back catalogue. Have a lovely two weeks and I’ll see you soon folks.
Outro
24:18-25:13
Ahoy team anaesthesia. You’ve survived yet another episode of Gas Gas Gas. Now if you found it useful or harrowingly awful please like and subscribe, drop us a star or 12 and follow with whichever podcast platform you find yourself using. Please leave a comment or ping off an email if you think I need to square something away. Now there are a bunch of ways to support the costs of Gas Gas Gas. From buying me a coffee to venturing forth via an affiliate link to the hoard of joyfully curated SBA questions from Teach Me Anaesthetics. Those links are on the website and in the show notes. Speaking of website, definitely check out gasgasgas.uk for the show notes, diagrams, details, the references. Now we all know guys that this is a bucket of content to consume and it is like drinking from a fire hose. So I want to finish by saying take it day by day. Don’t overcook yourself, don’t freak out and keep studying.
Enjoyed this? Review on Apple Podcasts Rate on Spotify
Support the show Help keep the lights on SBA question bank @ Teach Me Anaesthetics